Improved percutaneous endoscopic translaminar approach for lumbar foraminal stenosis at L5/S1
The operative approach for lumbar foraminal stenosis (LFS) is one of the most challenging for spinal surgeons.[1-3] Excessive removal of the dorsal area of the foramen in a posterior approach can easily lead to iatrogenic spondylolysis, subsequently increasing lumbar instability and spondylolisthesis. A posterolateral approach is one solution to this problem. However, LFS in the L5/S1 region is difficult to treat using a posterolateral approach, because of an anatomic peculiarity: the lateral aspect of the foramen is surrounded by the L5 transverse process, sacral ala, and hypertrophic facet joint. LFS usually develops after the age of 50 and is one of the most common degenerative spinal diseases in the elderly. As a result of this the vertebral height generally decreases, depending on the degree of disc degeneration. Compared with the younger patients without disc degeneration, the surgical access becomes more difficult.
Recent advances in the percutaneous endoscopic lumbar discectomy (PELD) technique have made access to the lateral aspect of the lumbar foramen possible without excessive removal of surrounding structures.[4] Despite this advance using a fully endoscopic system, access to the L5/S1 region remains difficult. At L5/S1, the surgeon cannot adequately tilt the endoscope to access the medial portion of the foramen due to the obstacle created by the ipsilateral iliac crest. To access the medial part of the L5 foramen, we improved the PELD approach, which had been developed for lumbar disc herniation (LDH) with migration into the hidden zone.[5]
This improvement uses a primarily posterior approach through an 8-mm skin incision placed just above the corresponding pars interarticularis. The endoscope sheath is placed on the surface, and the dorsal area of the foramen is removed with a high-speed drill.
The entrance keyhole is small enough to prevent iatrogenic spondylolysis; however, the area of bone removal is enlarged in the deep part of the hole [Figure 1]. Therefore, the dorsal part of the foramen is adequately removed. Bone removal and widening toward the bottom requires special skill with the 25 degree angled endoscope. Therefore, not only preservation of the pars interarticularis but also removal of the medial part of the foramen is accomplished using this percutaneous endoscopic translaminar approach (PETA) [Figure 1].
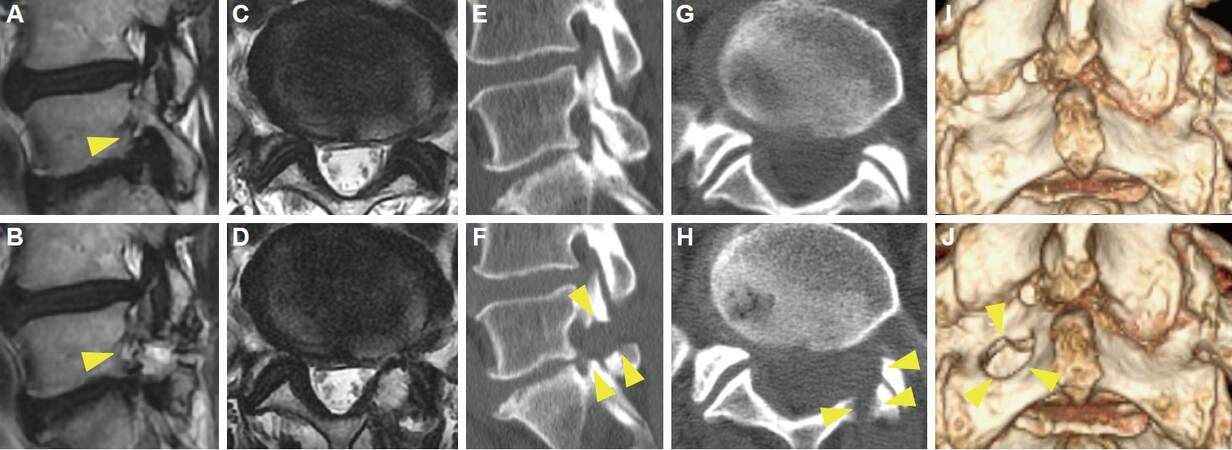
Figure 1. Representative case of improved PETA. A 70-year-old male complained of left leg pain that worsened with walking. Neurological examination revealed no muscle weakness and a negative SLR sign. Sagittal lumbar MRI revealed left foraminal stenosis at the L5/S1 intervertebral disc level, with marked compression of the left L5 nerve root (A, arrow head). We performed PETA, and his symptom improved (NRS 8→0, JOA 15→22) 2 weeks after PETA. Postoperative MRI revealed decompression of the foramen (B, arrow head). Comparison of preoperative (C, E, G, I) and postoperative (D, F, H, J) CT findings demonstrated the extent of bone removal (arrow heads). (A, B, E, F) sagittal view, (C, D, G, H) axial view, (I, J) 3-dimensional reconstruction. PETA: percutaneous endoscopic translaminar approach; SLR: straight leg rising; NRS: Numeric Rating Scale; JOA: Japanese Orthopedic Association; MRI: magnetic resonance imaging; CT: computed tomography
Previously, the combination of decompression of the foramen and a fusion procedure was performed in patients with significant LFS at L5/S1.[6] However, improvements in the decompression technique reported by several investigators make it possible to avoid fusion.
Among these improvements, intra-extracanal and contralateral interlaminar approaches show promise for the prevention of iatrogenic spondylolysis.[7,8] Although their common basis is in the operative direction of dorsomedial to ventrolateral decompression, these approaches are still invasive, and involve muscle retraction and extensive bone and ligament removal. The combination of a fully endoscopic system and development of a high-speed drill for use through a long and narrow endoscopic lumen has created options for minimally invasive spinal surgery for LFS at L5/S1 [Figure 2]. The improved PETA results in almost no damage to muscle and minimal removal of the surface of the vertebral arch.
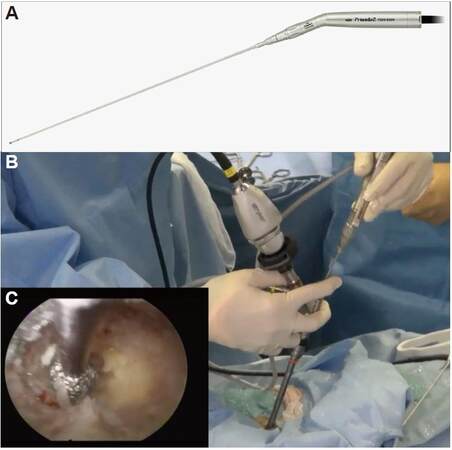
Figure 2. (A) Photograph of the electrical high-speed drill used for percutaneous endoscopic translaminar approach (NSK-Nakanishi Japan, Tokyo, Japan), and (B) intraoperative manipulation of the drill. The surgeon must hold both the endoscope and the drill. It will be necessary to develop a device to hold the endoscope to simplify this procedure. The surgeon can confirm the extent of bone removal in the endoscopic visual field (C)
PETA was first proposed by Dezawa et al.[5] for the treatment of LDH with migration into the hidden zone. The authors used PETA in 9 cases of hidden-zone LDH, and successful removal of LDH was confirmed using postoperative magnetic resonance imaging in all cases. They created a 4-mm bone hole using a high-speed drill with a diameter of 3.2 mm, as a larger bone hole is required for the treatment of LFS at L5/S1. We therefore made an approximately 10 mm diameter entry hole at the pars interarticularis using a high-speed drill with a diameter of 3.5 mm. Dezawa et al.[5] pointed out that the disadvantages of PETA are its technically demanding nature and the hand-eye coordination learning curve, and recommended that PETA should only be attempted after developing significant skill in standard endoscopic techniques. Du et al.[9] also used PETA in 7 highly down-migrated LDH cases and obtained good outcomes. Compared with the treatment of LDH, the treatment of LFS is more difficult, because LFS occurs in older patients with combined facet joint osteoarthritis and disc degeneration. Further refinement of the technique will make this improved PETA available to spine surgeons who treat such patients.
In general the duration of the operation for PETA exceeds 1 h (longer than that for PELD), we therefore perform PETA under general anesthesia. To avoid possible nerve damage, measurement of transcranial motor evoked potentials (Tc-MEPs) is performed. Tc-MEPs are recorded in tibialis anterior, extensor hallucis longus, and gastrocnemius muscles in the lower extremities as described in our previous report.[10] The start point to perform drilling is the most important matter for PETA to minimalize the bone removal. To determine appropriate position of drilling, antero-posterior (AP) and lateral views of a fluoroscope are used just after patient’s positioning on an operative bed (skin incision is placed at medial border of L5 pedicle on AP view and at caudal margin of the pedicle on lateral view). During the operation, we leave the fluoroscope across the center of the operative table in order to ensure appropriate drilling. The thickened superior articular process is carefully removed using the electrical high-speed drill. After removal of these hypertrophic tissues, we confirm good exposure and decompression of the corresponding nerve root by tilting and rotating the 25-degree angled endoscope. As a lens of camera is attached to the tip of the endoscope and enables to observe closely a wide range of the operative field. The extent of decompression is also confirmed with fluoroscopy using a dissector (width 3.0 mm). At the final stage of bone removal, we can easily confirm the tip of the dissector penetrating vertebral foramen. If a surgical navigation system for PETA will be developed, it can lead to considerably higher precision of bone removal.
With the aging of population and advances in diagnostic methods, the need for surgical treatment of LFS has increased.[6] On the other hand, patient demand for minimally invasive surgery is also rapidly increasing. The development of new equipment for fully endoscopic spinal surgery will be key to the introduction of this procedure. I expect that equipment manufacturers will partake in the development of this new surgical approach.
Financial support and sponsorship
None.
Conflicts of interest
There are no conflicts of interest.
Patient consent
All involved patients gave their consent forms.
Ethics approval
Not applicable.
REFERENCES
1. Pirris SM, Dhall S, Mummaneni PV, Kanter AS. Minimally invasive approach to extraforaminal disc herniations at the lumbosacral junction using an operating microscope: case series and review of the literature. Neurosurg Focus 2008;25:E10.
2. Epstein NE. Evaluation of varied surgical approaches used in the management of 170 far-lateral lumbar disc herniations: indications and results. J Neurosurg 1995;83:648-56.
3. Epstein NE. Foraminal and far lateral lumbar disc herniations: surgical alternatives and outcome measures. Spinal Cord 2002;40:491-500.
4. Yokosuka J, Oshima Y, Kaneko T, Takano Y, Inanam H, Koga H. Advantages and disadvantages of posterolateral approach for percutaneous endoscopic lumbar discectomy. J Spine Surg 2016;2:158-66.
5. Dezawa A, Mikami H, Sairyo K. Percutaneous endoscopic translaminar approach for herniated nucleus pulposus in the hidden zone of the lumbar spine. Asian J Endosc Surg 2012;5:200-3.
6. Orita S, Inage K, Eguchi Y, Kubota G, Aoki Y, Nakamura J, Matsuura Y, Furuya T, Koda M, Ohtori S. Lumbar foraminal stenosis, the hidden stenosis including at L5/S1. Eur J Orthop Surg Traumatol 2016;26:685-93.
7. Paolini S, Ciappetta P, Raco A, Missori P, Delfini R. Combined intra-extracanal approach to lumbosacral disc herniations with bi-radicular involvement. Technical considerations from a surgical series of 15 cases. Eur Spine J 2006;15:554-8.
8. Zekaj E, Menghetti C, Saleh C, Isidori A, Bona A, Aimar E, Servello D. Contralateral interlaminar approach for intraforaminal lumbar degenerative disease with special emphasis on L5-S1 level: a technical note. Surg Neurol Int 2016;7:88.
9. Du J, Tang X, Jing X, Li N, Wang Y, Zhang X. Outcomes of percutaneous endoscopic lumbar discectomy via a translaminar approach, especially for soft, highly down-migrated lumbar disc herniation. Osteoporos Int 2016;40:1247-52.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Koga H. Improved percutaneous endoscopic translaminar approach for lumbar foraminal stenosis at L5/S1. Mini-invasive Surg 2017;1:3-5. http://dx.doi.org/10.20517/2574-1225.2016.07
AMA Style
Koga H. Improved percutaneous endoscopic translaminar approach for lumbar foraminal stenosis at L5/S1. Mini-invasive Surgery. 2017; 1: 3-5. http://dx.doi.org/10.20517/2574-1225.2016.07
Chicago/Turabian Style
Koga, Hisashi. 2017. "Improved percutaneous endoscopic translaminar approach for lumbar foraminal stenosis at L5/S1" Mini-invasive Surgery. 1: 3-5. http://dx.doi.org/10.20517/2574-1225.2016.07
ACS Style
Koga, H. Improved percutaneous endoscopic translaminar approach for lumbar foraminal stenosis at L5/S1. Mini-invasive. Surg. 2017, 1, 3-5. http://dx.doi.org/10.20517/2574-1225.2016.07
About This Article
Copyright
Author Biographies

Data & Comments
Data
 Cite This Article 0 clicks
Cite This Article 0 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.