Modified parasternal approach is a good alternative for aortic valve surgery
Abstract
Aim: Cardiac surgery, as with other surgical specialties, has moved toward minimally invasive procedures. Currently, since the cardiopulmonary bypass machine remains necessary for most cardiac surgery procedures, efforts have focused on decreasing surgical trauma by limiting vascular access sites and any unnecessary dissection. This study presents the authors’ approach for less invasive valve surgery, which aimed to avoid a conventional midline sternotomy and reducing the length of incision.
Methods: For patients with aortic valve involvement, parasternal approach was the primary choice. A longitudinal 5-6 cm incision was made one fingerbreadth lateral to the sternal border. The 3rd rib was cut at the chondrosternal junction and bent into the right pleural cavity. After either central aortic or peripheral cannulation, all procedures were completed under surgeon’s direct vision and conventional instruments. The rib was reduced into position with a wire to offer stability and eliminate postoperative chest deformity.
Results: The authors’ experience of more than 500 cases with the minimally invasive approach showed that bypass time and ischemic time for parasternal valve surgery were compatible with to a full-sternotomy approach. In this series, postoperative ventilation time, blood product consumption and overall mortality were reduced.
Conclusion: Reviewing the parasternal aortic valve series of more than 500 cases, parasternal approach is safe, effective, and reproducible. The surgical trauma and blood product consumption were minimized with this approach. Multiple valve procedures and ablation for atrial fibrillation are also feasible. Stable sternoclavicular joints could facilitate early and aggressive activity of upper extremities for improved postoperative recovery. This approach could be a good alternative option in aortic or multiple valve surgical procedures.
Keywords
Introduction
Minimally invasive cardiac surgery (MICS) has been widely adopted.[1] Partial sternotomy for aortic valve replacement is the most common MICS for aortic valve replacement.[2] For multiple valve procedure, full sternotomy still remains the choice for most cardiac surgeons. There are several sternum-sparing approaches, such as the anterior thoracotomy,[3,4] lateral thoracotomy[5] and right parasternotomy.[6] Among these three approaches, we modify the Cosgrove’s parasternal approach to avoid the paradoxical chest movement and make it usable for most cardiac surgeons. In this series, improved cosmetic results, lower wound infection rates, shorter hospital and intensive care unit stay were found with the minimally invasive approach.
Methods
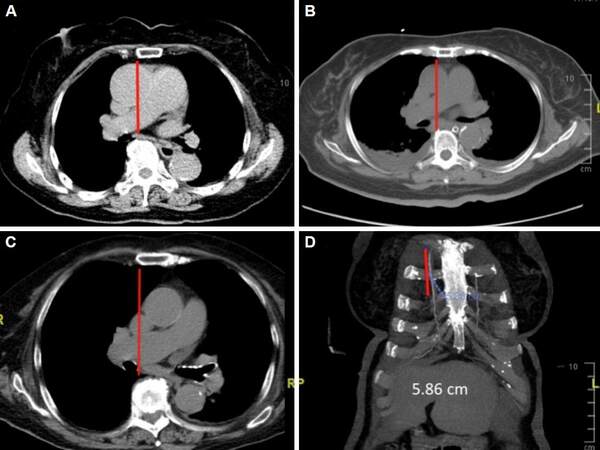
In 2003, our institution adopted the minimally invasive approach. Patients with isolated mitral procedures or mitral and tricuspid procedures were done through right lateral mini-thoracotomy. For patients with aortic valve involvement, Cosgrove’s idea[7] was adopted and modified with no rib resection. Initially, the two-rib approach was initiated with the following major selection criteria: adult single aortic valve cases, without chest wall deformity, without severe chronic obstructive pulmonary disease, and without aneurysmal or aortoiliac occlusive disease. Every single patient considered for this approach had pre-operative chest, abdomen and pelvis computed tomography (CT) scans to exclude aneurysmal aortic disease, dense calcifications, or occlusive aorto-iliac disease, which were considered to be contraindications to peripheral cannulation. In addition, the axial and frontal CT scans helped to identify the distance and relative position between skin incision and aortic annulus [Figure 1]. The patient was placed supine and intubated with double-lumen endotracheal tube for temporary one-lung ventilation during costo-chondral flap preparation. One pillow was put under right scapula, which tilted the patient left approximately 30 degrees. External defibrillation pads were applied routinely preoperatively.
Figure 1. (A-C) Cross-sectional views of ascending aorta. The red lines indicate the planes of right sternal borders. Axial CT scan revealed relative position of ascending aorta versus sternal border. Aortic valve exposure is less favorable from A to C. Frontal reconstruction of CT scan offers the estimation of longitudinal length; D: distance from the lower margin of second rib to the upper margin of fourth rib. Less than 4 cm would be less favorable for one-rib approach. CT: computed tomography
The skin incision was made longitudinally one fingerbreadth parallel to right lateral sternal border from the lower margin of 2nd rib to the upper margin of 5th rib. A subcutaneous pocket was created for better detachment of the underlying muscle. The pectoralis major muscle was cut in a reversed C shape from the sternal border attachment to allow lateral retraction naturally. The intercostal muscle was cut underneath the 2nd rib and above the 5th rib. The right internal mammary artery and veins were encountered at both intercostal spaces and carefully divided after clip ligation. The 3rd and 4th ribs were cut at sternochondral junction with a rib cutter. The sharp margin was trimmed with a bone file to prevent inadvertent lung injury. The myocostal flap was bent into right pleural cavity after manual fracture. The fracture point was adjusted based on the width needed for surgical exposure, which was approximately at the junction between rib and costal cartilage. Care was taken to maintain the periosteum and soft tissue attachment to ensure the bony healing and future wound stability. Then, a small-sized rib or sternal spreader (retractor) was inserted to keep constant exposure. The two blades of sternal or rib retractor could be asymmetric. The short blade was good for the sternal side and the longer blade was suitable for the lateral myocostal flap. Pericardium was opened longitudinally after removing the epicardial fat pad. Stay sutures were placed to bring the mediastinum right. Placement of the sternal retractor inside the pericardial cradle provided adequate exposure. If the ascending aortic exposure was adequate enough to accommodate aortic cannulation, crossclamp, cardioplegic needle, aortotomy and prosthetic manipulation, central cannulation could be performed. Otherwise, peripheral cannulation, mainly femoral and occasionally axillary cannulations were used. Femoral vein cannulation was routinely utilized to ensure better exposure under limited skin incision. Wire-reinforced aortic cannula was chosen for better positioning without the risk of kinking. Single femoral venous cannulation plus vacuum assisted venous drainage usually provided adequate drainage. From our experience, left neck central venous line was preferred. Right neck was preserved for surgeon manipulation. Echo-guided 4-Fr introducer sheath was inserted via right interal jugular vein before commencement of cardiopulmonary bypass (CPB). This strategy was helpful when inadequate venous drainage was encountered in order to insert a superior vena cana cannula or a tranvenous pacemaker wire when needed.
After commencing CPB, the aortopulmonary window was dissected to facilitate ascending aorta looping using vascular tape. This step was helpful not only for securely cross clamping the aorta but also for adding sutures for hemostasis at both ends of aortotomy. A malleable left ventricular venting catheter was inserted via the right upper pulmonary vein through mitral valve into left ventricle. It was helpful to maintain constant bloodless exposure of aortic valve, to decompress left ventricle and to de-air after crossclamp was removed and before CPB was weaned off.
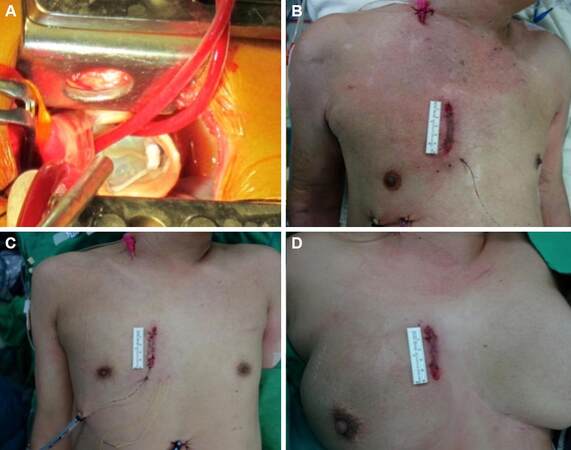
Using the traction sutures on the pericardium and fixing the cross clamp rightward, the aortic exposure was usually adequate enough for having an aortotomy and delivering cardioplegic solution, even with coronary ostia balloon catheters. Aortic leaflets and annulus could be well-exposed for inspection. Decalcification and trimming of leaflets were then performed meticulously. Repair or replacement of the aortic valve was done by conventional techniques and instruments [Figure 2A]. For mitral valve procedures, three approaches could be chosen according to patient’s anatomy and complexity of mitral procedures. By using femoral bicaval cannula and snaring of both superior and inferior vena cava, trans-septal approach offered the best mitral exposure. Through Waterston’s groove, the trans-lateral approach was a viable alternative, but the mitral valve would be farther away than trans-septal exposure. For a simplified suture annuloplasty, the left atrial dome approach was an attractive option. Meanwhile, tricuspid valve procedures could be straightforwardly done. Ablation for atrial fibrillation (AF) could be performed both endocardially and epicardially by cryoprobe or unipolar radiofrequency probe.[8] Closure of left atrial appendage was feasible. In brief, most valvular procedures could be achieved through our two-rib parasternal approach, except for pulmonary valve procedures. If temporary ventricular pacemaker wires were considered, insertion under total decompression of heart chambers over right ventricle was highly recommended. Transvenous pacemaker wire through right internal jugular or right subclavian vein approach was an alternative.
Figure 2. (A) Surgical exposure revealed the well-seated bioprosthesis and two coronary ostia balloon catheters for cardioplegic solution delivery; (B-D) the other three pictures showed the final skin closure immediately after the completion of surgery
After our first hundred cases, a single-lumen endotracheal intubation was employed to simplify anesthetic induction process. Temporary cessation of mechanical ventilation would suffice for pleural entry and bending of the myocostal flap. This approach ensured the same prep as a conventional sternal one, except for left-tilted position and external defibrillation pads.
For the next stage of our experience, we moved to one-rib parasternal approach. All surgical details were similar, except for a smaller incision and only the 3rd rib was cut. A one-rib approach was usually aimed for an isolated aortic procedure. The major determinant for this approach would be the length between the lower margin of 2nd rib and upper margin of 4th rib. For patients with a narrow 2nd intercostal space, this would be difficult to proceed [Figure 1D]. For more than two thirds of these patients, central aortic cannulation could be achieved. Some patients had concomitant mitral repair as well. We also developed epicardialcryo-ablation for AF using pulmonary vein isolation through this limited incision.
After the completion of valvular procedures, CPB was weaned off and hemostasis was achieved. Two Jackson-Pratt drains were inserted into pericardial and pleural space, respectively. Pericardium was closed with interrupted sutures. The myocostal flap was reduced back into the anatomical position. Any partially broken ribs was fixed with a pediatric wire to sternum. For patients with widened 2nd intercostal space, several interrupted, braided, non-absorbable sutures were used as a fence to divide the space and prevent lung herniation. Mesh for abdominal hernia and ePTFE patch had been placed in selective cases. The pectoral major muscle was brought back centrally and fixed with interrupted sutures. Subcutaneous fascia and skin were closed [Figure 2B-D]. Our postoperative pain management consisted of controlled local anesthetic infusion and a patient-controlled analgesia pump.
Results
From 2004 to 2016, 543 parasternal cardiac operations were performed at our institution. The cases included 297 isolated aortic valve, 124 aortic and mitral, 45 aortic, mitral and tricuspid procedures and miscellaneous applications. Nine percent were redo procedures. Average time for parasternal cross clamp, CPB, operation were 61.84, 101.43 and 243.00 min, respectively [Table 1]. Average ventilation time and intensive care unit stay were 13 h and 2.4 days [Table 2]. Surgical mortality was 1.9%. There was one conversion to sternotomy for persistent bleeding. Five patients had perioperative central nervous complications. Retrograde flow from femoral cannulation and inadequate de-airing were considered to be potential causes. Parasternal wound complications were rare and self-limited. The well vascularized pectoralis muscle coverage potentially decreased local infection. Eight patients had a wound infection that required additional limited surgical debridement without the entry of mediastinum. Three of them had local infection related to temporary pacemaker wires [Table 3]. Pain analogue scale was usually less than 2-3 under our aggressive pain management. More than 90% patients were satisfied with their operative wounds.
Demographic data of the parasternotomy patient group
| Charecteristics | Total (n = 543) | Male (n = 339) | Female (n = 204) |
|---|---|---|---|
| Age, years | |||
| Average | 62.63 ± 14.00 | 61.58 ± 14.20 | 64.39 ± 13.50 |
| Median | 64 | 62 | 65 |
| BMI | 24.22 | 24.56 | 23.66 |
| Euro score | |||
| < 1 | 70 | 54 | 16 |
| 1-5 | 230 | 136 | 94 |
| 6-10 | 57 | 28 | 29 |
| 11-20 | 29 | 18 | 11 |
| > 20 | 12 | 7 | 5 |
| Concomitant disease, n (%) | |||
| Diabetes | 88 (16.2) | 51 (15.0) | 37 (18.1) |
| Dyslipidemia | 124 (22.8) | 74 (21.8) | 50 (24.5) |
| Hypertension | 279 (51.4) | 172 (50.7) | 104 (51.0) |
| COPD | 46 (8.5) | 33 (9.7) | 13 (6.4) |
| Status of the procedure | |||
| Elective | 536 | 335 | 201 |
| Urgent | 2 | 2 | 0 |
| Emergent | 5 | 2 | 3 |
| Cardiac surgery | |||
| First | 501 | 313 | 188 |
| Second | 35 | 22 | 13 |
| Third | 7 | 4 | 3 |
| Fourth or more | 0 | 0 | 0 |
| LVEF distribution | |||
| LV dysfunction poor or LVEF < 30% | 25 | 17 | 8 |
| LV dysfunction moderate or LVEF 30-50% | 99 | 70 | 29 |
Operative and hospital data
| Charecteristics | Para (n = 543) | Full (n = 2,991) | P |
|---|---|---|---|
| Hospital stay (days) | 14.18 ± 8.17 | 20.55 ± 19.58 | < 0.001 |
| Intensive care unit stay (h) | 57.54 ± 23.70 | 131.49 ± 197.90 | < 0.001 |
| Cross clamp time (min) | 61.84 ± 25.60 | 71.10 ± 46.63 | < 0.001 |
| Perfusion time (min) | 101.43 ± 41.72 | 135.78 ± 78.41 | < 0.001 |
| Operative time (min) | 243.73 ± 68.77 | 283.14 ± 114.85 | < 0.001 |
| Ventilation time (h) | 13.77 ± 13.90 | 75.73 ± 202.72 | < 0.001 |
Complication rate, n (%)
| Complications | Para (n = 543) | Full (n = 2,991) |
|---|---|---|
| Wound infection | 3 (0.6) | 37 (1.2) |
| Stroke | 3 (0.6) | 116 (3.9) |
| Renal failure* | 11 (2.0) | 186 (6.2) |
No iatrogenic and retrograde aortic dissections or phrenic nerve injuries were identified. Groin complications like seroma were common, although the majority were self-limited. In the latter half of patients, a self-closure device and echo-guided femoral artery puncture were employed to minimize groin complications significantly. Lung complications were rare. Some patients develop right hilar haziness in the first few days which might result from local compression of myocostal flap during the operation. However, lung recruitment by pressure controlled lung expansion during wound closure helped to improve atelectasis and facilitate early extubation. No evidence of non-union, pseudojoint, or chest cage deformity were identified. Any broken ribs were fixed with pediatric wire and lateral soft tissue attachment. Paradoxical movement and lung herniation were rare and typically happened at the 2nd intercostal space. This was well-controlled since interrupted sutures were used for 2nd intercostal space. Local dimpling due to pectoralis muscle atrophy occurred in our series. Therefore, an inverted C incision for pectoralis muscle instead of cutting straightforward muscle following skin incision was performed. Upper extremity exercise was also encouraged after patients were transferred out of intensive care unit. Instructions were given to patients on how to protect the wound. Simple one-hand compression over the wound offers stability and pain reduction, especially while coughing. Two or even one small Jackson-Pratt drains rather than chest tubes also made early ambulation easier.
Discussion
MICS is a growing field with the goals to eliminate cardiopulmonary bypass and to avoid a sternotomy. Among the three sternum-sparing approaches, anterior thoracotomy gains the most acceptance especially in Europe when dealing with single aortic valve replacement. It takes advantage of the anatomic proximity to aorta. However, the elliptical spread of intercostal space and its perpendicularity to the ascending aorta limits the exposure. Even though the 3rd rib is transected at sternochondral junction, it still provides limited access to the aortic valve annulus.[9] Skin incision of lateral thoracotomy is usually made close to axilla and is intended to be hided under anterior axillary fold. The incision is the largest among these three approaches. Sizable wounds of the intercostal muscles need to be cut in order to gain better exposure. Besides, extended-length instruments and knot-pusher are required to finish the valve replacement.[5] This is only reserved for most experienced and highly skilled surgeons. Here, we share our experience of the parasternal approach.
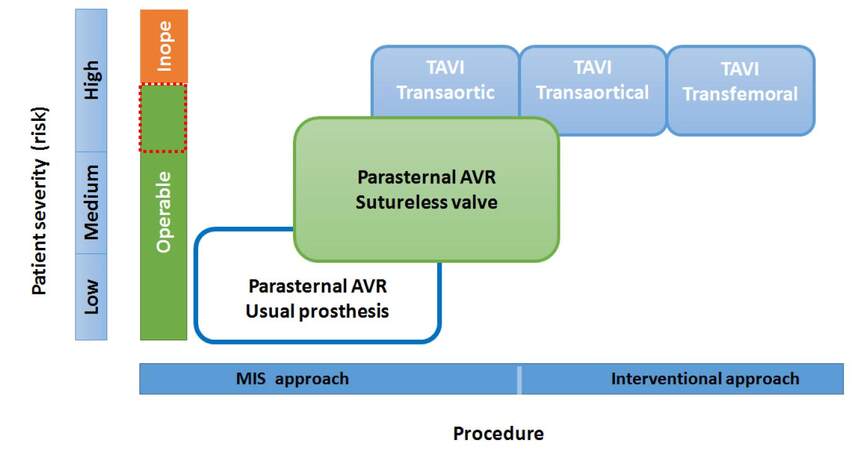
The treatment of aortic valve diseases are based on patients’ risk categories and invasiveness or complexity of procedures [Figure 3]. The original parasternal approach developed by Cosgrove was aimed for a sternum-sparing purpose. Although the take-down of costal cartilages facilitated wide exposure, however, it resulted in anterior chest deformity, lung herniation, and paradoxical movement. Cosmetic results and patients’ satisfaction may not be attained.[10] In our series, these adverse outcomes were significantly improved.
Figure 3. Our surgical strategy for aortic valve procedures were mainly categorized by patients' risk severity and the complexity/invasiveness of procedures. MIS: minimally invasive surgery; AVR: aortic valve replacement; TAVI: transcatheter aortic valve implantation
For surgeons considering to employ our parasternal approach, they should be familiar with conventional aortic valve surgery and have a certain amount of experience independently. Team members’ orientation is usually simple. No additional effort is needed from anesthesiologists. Operative nurses and surgical assistants are well instructed in advance since they may not be able to view the details through limited exposure. Head-mounted camera from the surgeon’s headlight would be helpful to keep crews on the same page and also for educational purpose. Extended length instruments are only needed for deeply seated mitral valves. There is limited or even no additional investment.
For patients who are considered for our parasternal approach, correct diagnoses are crucial. Intention for surgical intervention should be limited to mainly aortic valve procedures. Associated procedures, like mitral valve, tricuspid valve, and AF ablation could be considered following the progress of learning curve. Physical examination is important. The width of 2nd, 3rd and 4th intercostal space tells us the potential working space [Figure 1D]. Chest wall deformities, like pigeon chest or funnel chest, and narrow intercostal space may not be suitable. Preoperative images are extremely helpful for defining the accessibility or searching the contraindications. Absolute contraindications would be defined as poor exposure which precludes the completion of proposed procedures, complex procedures in early learning process, and ascending aortic or arch aneurysm.
Chest films help us to have ideas of proximity of proposed incision and aorta. However, CT scan from neck to pelvis offers the most information.[11] Contrast medium is usually not required, especially for patients proposed to have operations within 48 h.[12] Aortic aneurysm and aorto-iliac disease are contraindications for peripheral cannulation, although thigh-brachial index or ankle-brachial index could offer the extent of severity. However, central aortic cannulation is usually possible and axillary artery cannulation could be an alternative if inadequate exposure is a concern. Elevated stroke risk is always a concern for certain patients with MICS using femoral arterial cannulation.[13,14]
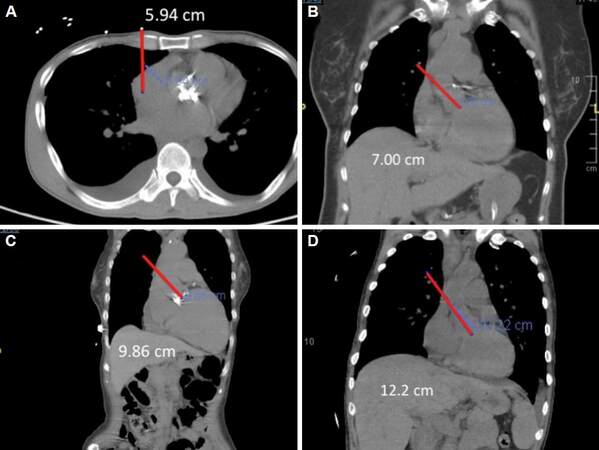
The location of the ascending aorta in relation to sternum is crucial. More than half of ascending aortic circumference are located right lateral to right sternal border.[15] The measurement from the center of proposed skin incision, usually 2 cm right lateral to sternum border over the 3rd rib, to aortic annulus is very helpful. This is a three-dimensional distance consisting of the root of square sum of three parameters which could be acquired from both axial and frontal reconstruction of CT scan [Figure 4]. However, it tends to be closer due to the difference between full inspiration during CT scanning and full muscle relaxation during operation. The distance in between 6 to 12 cm would be acknowledged as friendly for our parasternal approach. Space for all ascending aortic interventions should be considered, including central aortic cannulation, cross clamp, cardioplegic or de-airing needle and reasonable room for aortotomy and prosthesis manipulation. Shorter distance usually indicates limited or shorter exposure of ascending aorta which necessitates peripheral cannulation and very low aortotomy. Farer distance clearly indicates difficulty in exposure, suturing and knot tying. Exposure may be acceptable after commencement of CPB and aortic cross-clamp. However, care should be given for meticulous suturing and hemostasis for aortotomy, especially at both ends. For those with limited exposure, additional stitches for overt bleeding may not be easy once CPB is weaned off and the heart is fully loaded. Complex procedures, like remodeling procedure, reimplantation procedure, Bentall operation, annular enlargement, peri-annular abscess patch repair for infective endocarditis and redo aortic valve replacement have been performed using two-rib parasternal approach in our series. Hemostatic products would be helpful for those suture lines without second chance of hemostasis.
Figure 4. The three-dimensional distance consisting of the root of square sum of simplified two parameters (A: depth from skin to the level of annulus; B-D: oblique distance from the projection of 3rd rib to annulus) which could be acquired from both axial and frontal reconstruction of computed tomography scan. The distance offers surgeons the feasibility and difficulty of this approach
Compared with anterior thoracotomy, similarly, both approaches may need peripheral CPB and have divided or torn right internal mammary artery, broken costal cartilage, lower conversion rate, and limited lung herniation. The advantage of “so-called” intact chest cage often counteracts by its limited exposure. Therefore, its application is limited to highly selected cases. Most surgeons prefer upper partial sternotomy rather than anterior thoracotomy. Conversely, the parasternal approach offers better exposure to the aortic root, annulus, and is technically easier for beginners initiating sternum-sparing approaches. It is more anatomically oriented and could be a good alternative for aortic valve surgery, especially for patients with the need for better exposure. Additionally, this approach offers opportunity for multi-valve surgery (including mitral and tricuspid).
Our parasternal approach carries additional benefits. Wound complications are rare, mainly due to the stability and muscle flap coverage. It maintains the integrity of sternum, manubrium, and sternoclavicular joints. From anatomical experience, upper extremities (UE) are connected to axial skeleton through clavicles, sternoclavicular joints and chest cage. Our approach reduces pain from UE movement. Early return of full range of UE motility improves post-operative lung function and quality of life.
With the availability of sutureless aortic prostheses, the aortic valve replacement could be facilitated.[16,17] High transverse aortotomy is easily achieved. Decalcification of the annulus and nadir sutures could be easily accomplished. The parasternal approach offers a versatile platform for wide-range of aortic valve procedures. After we developed this technique, it has become our standard practice and primary choice for aortic valve surgery. Our propensity-adjusted analysis shows that the parasternal approach may be a good minimally invasive alternative to full sternotomy for cardiac valve operations.[6]
In conclusion, improved cosmetic results, less pain and anagelsic use, and faster recovery have been reported. It has been generally well-accepted by patients and also by surgeons performing minimally invasive aortic valve replacement.[18] We agree that full sternotomy procedures have achieved excellent surgical outcomes. It may not be easy to elaborate the differences in outcomes simply through various surgical incisions. Also, new approaches, if not been well conducted, could be detrimental due to unfamiliar and unexpected complications. Open mindedness and curiosity, on-site case observations, and proctor guided practices by well-experienced surgeons are key elements in adopting new approaches that can offer true benefits to selected patients. Preoperative images help us to plan carefully and avoid potential complications. Starting with simple, isolated and straightforward procedures is always the rule of thumb to negotiate the learning curve of new techniques. From our experience, our parasternal approach is an easily learned and performed procedure.[6] Our long-term follow up also proves that this a durable procedure.
Authors’ contributions
K.M. Chiu contributed solely to this article.
Financial support and sponsorship
None.
Conflicts of interest
There are no conflicts of interest.
Patient consent
Patients’ informed consent were obtained before operations.
Ethics approval
This study is waived for ethical approval from the author’s institution review board.
REFERENCES
1. Navia JN, Cosgrove DM 3rd. Minimally invasive mitral valve operations. Ann Thorac Surg 1996;62:1542-4.
2. Brown ML, McKellar SH, Sundt TM, Schaff HV. Ministernotomy versus conventional sternotomy for aortic valve replacement: a systematic review and meta-analysis. J Thorac Cardiovasc Surg 2009;137:670-9.
3. Bowdish ME, Hui DS, Cleveland JD, Mack WJ, Sinha R, Ranjan R, Cohen RG, Baker CJ, Cunningham MJ, Barr ML, Starnes VA. A comparison of aortic valve replacement via an anterior right minithoracotomy with standard sternotomy: a propensity score analysis of 492 patients. Eur J Cardiothorac Surg 2016;49:456-63.
4. Rodriguez E, Malaisrie SC, Mehall JR, Moore M, Salemi A, Ailawadi G, Gunnarsson C, Ward AF, Grossi EA; Economic Workgroup on Valvular Surgery. Right anterior thoracotomy aortic valve replacement is associated with less cost than sternotomy-based approaches: a multi-institution analysis of 'real world' data. J Med Econ 2014;12:846-52.
5. Ito T, Maekawa A, Hoshino S, Hayashi Y. Right infra-axillary mini-thoracotomy for aortic valve replacement. Ann Cardiothorac Surg 2015;4:78-80.
6. Chiu KM, Chen RJ, LinTY, Chen JS, Huang JH, Huang CY, Chu SH. Right mini-parasternotomy may be a good minimally invasive alternative to full sternotomy for cardiac valve operations-a propensity-adjusted analysis. J Cardiovasc Surg 2016;57:111-20.
7. Cosgrove DM 3rd, Sabik JF. Minimally invasive approach for aortic valve operations. Ann Thorac Surg 1996;62:596-7.
8. Gillinov AM, Gelijns AC, Parides MK, DeRose JJ Jr, Moskowitz AJ, Voisine P, Ailawadi G, Bouchard D, Smith PK, Mack MJ, Acker MA, Mullen JC, Rose EA, Chang HL, Puskas JD, Couderc JP, Gardner TJ, Varghese R, Horvath KA, Bolling SF, Michler RE, Geller NL, Ascheim DD, Miller MA, Bagiella E, Moquete EG, Williams P, Taddei-Peters WC, O'Gara PT, Blackstone EH, Argenziano M; CTSN Investigators. Surgical ablation of atrial fibrillation during mitral-valve surgery. N Engl J Med 2015;372:1309-409.
9. Glauber M, Gilmanov D, Farneti PA, Kallushi E, Miceli A, Chiaramonti F, Murzi M, Solinas M. Right anterior minithoracotomy for aortic valve replacement: 10-year experience of a single center. J Thorac Cardiovasc Surg 2015;150:548-56.
10. Johnston DR, Roselli EE. Minimally invasive aortic valve surgery: Cleveland Clinic experience. Ann Cardiothorac Surg 2015;4:140-7.
11. Youssef SJ, Millan JA, Youssef GM, Earnheart A, Lehr EJ, Barnhart GR. The role of computed tomography angiography in patients undergoing evaluation for minimally invasive cardiac surgery: an early program experience. Innovations (Phila) 2015;10:33-8.
12. Di Tomasso N, Monaco F, Landoni G. Renal protection in cardiovascular surgery. F1000Res 2016;5:331.
13. Bedeir K, Reardon M, Ramchandani M, Singh K, Ramlawi B. Elevated stroke risk associated with femoral artery cannulation during mitral valve surgery. Semin Thorac Cardiovasc Surg 2015;27:97-103.
14. Lamelas J, Williams RF, Mawad M, LaPietra A. Complications associated with femoral cannulation during minimally invasive cardiac surgery. Ann Thorac Surg 2017;103:1927-32.
15. Glauber M, Ferrarini M, Miceli A. Minimally invasive aortic valve surgery: state of the art and future directions. Ann Cardiothorac Surg 2015;4:26-32.
16. Miceli A, Santarpino G, Pfeiffer S, Murzi M, Gilmanov D, Concistré G, Quaini E, Solinas M, Fischlein T, Glauber M. Minimally invasive aortic valve replacement with perceval S sutureless valve: early outcomes and one-year survival from two European centers. J Thorac Cardiovasc Surg 2014;148:2838-43.
17. Borger MA, Moustafine V, Conradi L, Knosalla C, Richter M, Merk DR, Doenst T, Hammerschmidt R, Treede H, Dohmen P, Strauch JT. A randomized multicenter trial of minimally invasive rapid deployment versus conventional full sternotomy aortic valve replacement. Ann Thorac Surg 2015;99:17-25.
18. Malaisrie SC, Barnhart GR, Farivar SA, Mehall J, Hummel B, Rodriguez E, Anderson M, Lewis C, Hargrove C, Ailawadi G, Goldman S, Khan J, Moront M, Grossi E, Roselli EE, Agnihotri A, Mack MJ, Smith JM, Thourani VH, Duhay FG, Kocis MT, Ryan WH. Current era minimally invasive aortic valve replacement: techniques and practice. J Thorac Cardiovasc Surg 2014;147:6-14.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Chiu KM. Modified parasternal approach is a good alternative for aortic valve surgery. Mini-invasive Surg 2017;1:81-8. http://dx.doi.org/10.20517/2574-1225.2017.03
AMA Style
Chiu KM. Modified parasternal approach is a good alternative for aortic valve surgery. Mini-invasive Surgery. 2017; 1: 81-8. http://dx.doi.org/10.20517/2574-1225.2017.03
Chicago/Turabian Style
Chiu, Kuan-Ming. 2017. "Modified parasternal approach is a good alternative for aortic valve surgery" Mini-invasive Surgery. 1: 81-8. http://dx.doi.org/10.20517/2574-1225.2017.03
ACS Style
Chiu, K.M. Modified parasternal approach is a good alternative for aortic valve surgery. Mini-invasive. Surg. 2017, 1, 81-8. http://dx.doi.org/10.20517/2574-1225.2017.03
About This Article
Copyright
Author Biographies

Data & Comments
Data

 Cite This Article 1 clicks
Cite This Article 1 clicks










Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.