Minimally invasive contact X-ray brachytherapy as an alternative option in patients with rectal cancer not suitable for bespoke surgical resection
Abstract
Surgery remains the gold standard treatment for rectal cancer. All published guidelines and most protocols recommend surgery as the standard of care. However, non-surgical management of rectal cancer is increasingly gaining acceptance as it avoids extirpative surgery and a stoma. In patients who are not suitable for surgery because of advancing age or medical comorbidities, and also in a small number of patients who are stoma phobic and refuse surgery, we need to consider an alternative treatment option to bespoke surgery. External beam radiotherapy is usually offered as an alternative. However, local regrowth rate is high and contact X-ray brachytherapy (Papillon treatment) boost can be added to reduce the risk of local regrowth after external beam radiotherapy. Case selection is important to achieve the best results.
Keywords
Introduction
Surgery remains the gold standard treatment for rectal cancer: all published guidelines and most protocols recommend surgery as the standard of care[1,2]. Selection of the best option for care is based on recommendations made during multidisciplinary team (MDT) meetings, which are now mandated in most countries to discuss treatment for all patients with rectal cancer. The majority of MDTs still recommend surgery even for early rectal cancer as this is regarded as the standard of care. However, non-surgical management of rectal cancer is increasingly gaining acceptance as it avoids extirpative surgery and a stoma[3]. In patients who are not suitable for surgery because of advancing age or medical comorbidities, and also in a small number of patients who are stoma phobic and refuse surgery, we need to consider an alternative treatment option to surgery[4]. In most cases, external beam radiotherapy alone (EBRT) or with chemotherapy (EBCRT) is offered as an alternative to surgery. It is likely that with EBCRT alone, 10%-30% of patients can achieve clinical complete response (cCR)[5,6]. For these patients, a “watch and wait” strategy can be offered that avoids extirpative surgery with a stoma. The published evidence indicates that in 25%-38% of cases, local regrowth can develop late after achieving a cCR following EBCRT alone[3,5]. In patients who are fit and agree to proceed, these recurrences require salvage surgery. Once the patient develops a regrowth, if they are not fit for surgery or if the patient refuse surgery, palliative care is the only available option and the majority will die from symptomatic progressive local regrowth. The burden of care for these patients can put strain on their health care providers as these patients can survive for months or even years. The alternative approach is to offer them contact X-ray brachytherapy (CXB, Papillon treatment) which can reduce the risk of local regrowth[4]. Case selection is important to achieve the best results.
Case selection for treatment
In patients who are not suitable for surgery, or in younger, medically fit patients who vehemently refuse surgery because of stoma phobia, an alternative treatment option is radical radiotherapy. There are two types of radiation: either external beam radiotherapy (EBCRT/EBRT) or CXB (using a Papillon).
The choice of radiation type and which treatment modality to start depends on: (1) stage of the tumor (cT1 ); (2) possible lymph node spread (cT2, cT3); and (3) size of the tumor ( < 3 cm or > 3 cm).
Inclusion criteria for CXB alone for early rectal tumors with curative intent
(1) mobile exophytic early rectal cancer (cT1);
(2) well to moderately differentiated adenocarcinoma;
(3) tumor size < 3 cm;
(4) no evidence of suspicious lymph nodes;
(5) no evidence of distant metastases;
(6) tumor within 12 cm of the anal verge;
(7) patient suitable for long-term follow-up.
Exclusion criteria
(1) poorly differentiated adenocarcinoma;
(2) presence of lymphatic or vascular invasion;
(3) bulky rectal cancer involving more than half the circumference ( > 3 cm);
(4) fixed rectal adenocarcinoma with deep ulceration (cT3, cT4).
Treatment strategies
Early small rectal cancers (cT1, cN0, < 3 cm)
When an asymptomatic early (cT1) small ( < 3 cm) rectal cancer is diagnosed (which usually occurs through the national bowel cancer screening program), the standard of care is to offer the patient surgery that may involve abdominoperineal resection of the rectum (APER) if the tumor is low in the rectum ( < 6 cm from the anal verge). If the patient is not suitable for surgery or refuses surgery, an alternative option is to offer them CXB (Papillon) alone[4].
More advanced larger rectal cancers (cT2, cT3a/cNo/cN1, > 3 cm)
If the tumor size is > 3 cm or if the tumor is at stage cT2 or cT3a, then the risk of lymph node metastases can be as high as 20%-30%. CXB alone is not suitable as the low energy X-rays have limited penetration that will not reach the lymph nodes in the meso-rectum. The usual standard of care is to offer these patients surgery[1,2]. However, if the patient is not suitable for surgery or refuses it, they can be offered an alternative treatment using external beam radiotherapy with or without chemotherapy. If the patient is fit, external beam chemo-radiotherapy, with a total dose of 45 Gy in 25 fractions over 5 weeks, or a biologically equivalent dose, can down-size the tumor. There is published evidence that in radio-responsive tumors[6], the malignant tissue is not just down-sized, but usually down-staged to either ypT0 or ypT1 as well. If the patient is not fit enough for this treatment or has a poor renal function, a short course of radiation (25 Gy in 5 fractions over 5 days) can be offered, with consideration of performing CXB boost after 4-6 weeks to improve local control[4,7].
Techniques
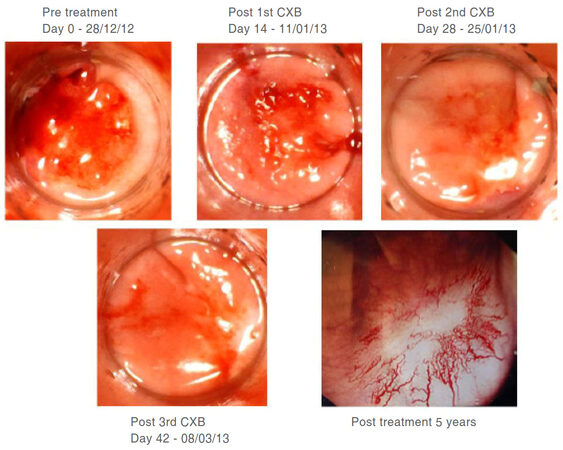
CXB uses a high dose (90 Gy) of low energy (50 KVp) X-rays which are targeted directly on the tumor under visual guidance. There are two machines currently available for CXB cancer therapy. First, the Papillon + X-ray brachytherapy unit is currently marketed by the British company Ariane Medical Systems, Ltd (Alfreton, UK). Additionally, the Xoft® Axxent® Electronic Brachytherapy System® (iCAD, Inc., San Jose, CA, USA) is currently only approved for breast, skin and gynecological cancers, but it is undergoing development for treatment of rectal neoplasms. The radiation dose applied at each treatment is quite high (30 Gy) but because the radiation energy is low (50 kV) and applied directly to the tumor in a small volume ( < 5 cc), the collateral damage to the normal surrounding tissues is limited. The treatment is given three times (30 Gy X 3) every two weeks. This regimen allows the normal tissues to recover during the 2-week break. There are three applicator sizes available: 30, 25 and 20 mm. The choice of applicator size depends on the tumor size which should be less than 30 mm (if the tumor size is > 30 mm, then EBCRT or EBRT is offered initially to down-size the tumor before CXB). The tumor is treated with a margin of 5 mm. In a responsive tumor, the lesion usually regresses centripetally [Figure 1], beginning immediately after the first fraction but mostly after the second fraction as illustrated in our case study[4,7].
Figure 1. Treatment response to contact X-ray brachytherapy (CXB). Case 1: 65-year-old male diagnosed with low rectal adenocarcinoma staged as cT1cN0cM0 on MRI and CT scan. Refused surgery including trans-anal endoscopic microscopic surgery (TEMS) and external beam radiotherapy. Patient’s choice. Treated only with CXB. Started his treatment in December 2012 after informed consent. Fully understand and accepted that CXB is non standard treatment for rectal cancer. Significant regression of tumour after only one fraction of CXB and no palpable or visible tumour after 2nd fraction. Clinical complete response (cCR) maintained after 5 years with good quality of life and bowel control. No bleeding despite being on clopidrogel
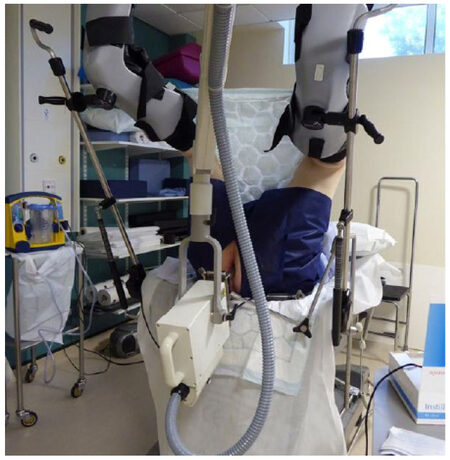
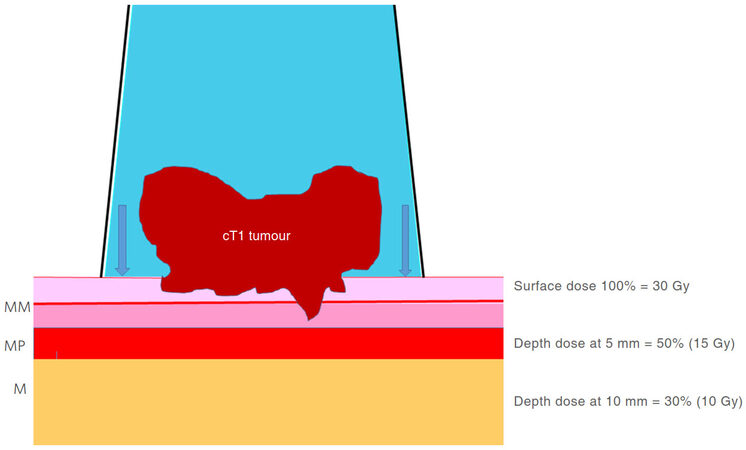
The treatment can be given as a day patient as the whole procedure usually takes less than 30 min. This includes the initial assessment with endoscopy and the treatment time is less than 150 s. The patient can be treated supine or prone, in a knee-chest position [Figure 2]. A rigid sigmoidoscope is inserted to assess the tumor size, position, and to select the size of the rectal applicator. Then the radiation is applied using a suitable rectal treatment applicator. The radiation dose of 30 Gy is delivered to the surface of the normal surrounding rectal mucosa. Therefore, exophytic lesions which protrude into the treatment applicator receive a much higher dose of radiation than 30 Gy at the surface of the tumor. In a radio-responsive tumor, the treated layer is shaved off after each radiation treatment until the tumor regresses completely to the base of the bowel wall, and finally is flush with the surface of the surrounding normal rectal mucosa. The deeper layers then get treated with subsequent fractions. At a depth of 5 mm below the surface of the rectal mucosa, where the muscularis propria (deep muscle) of the rectal wall is situated, the dose of CXB is reduced to 50% of the surface (applied) dose, and at a 10 mm depth, the dose is attenuated to 30% of the surface dose [Figure 3]. There is published evidence that 98% of the residual tumor is usually confined within the muscularis propria (5 mm deep from the rectal mucosa) for early stages (cT1, cT2) of rectal tumors[6]. We normally offer CXB boost treatments 4-6 weeks following EBCRT. However, if the residual tumor following EBCRT is still bulky and infiltrates more than 5 mm below the rectal mucosa (beyond the rectal wall full thickness) we can delay the treatment by few more weeks to see if there is further regression of the residual tumor before proceeding with the CXB boost.
Figure 3. Treatment diagram and depth dose. MM: muscularis mucosa; MP: muscularis propria; M: mesorectum
Follow-up
The risk of local neoplastic regrowth is usually highest within the first 2 years[4,7] and close follow-up is important during this period. Most of the regrowth is intraluminal[8] and can be detected by endoscopic examination, which should be carried out every 3 months during the first year, every 3-4 months during the second year, and every 6 months from the third to the fifth post-treatment year. Full colonoscopy should be done at 5 years if not performed earlier. Usually a digital rectal examination (DRE) is carried out just before inserting the endoscope for this procedure to assess any palpable local regrowth and its mobility. High-resolution whole-pelvis magnetic resonance imaging (MRI) should be done every 3-4 months during the first 2 years and at 6-month intervals in the third year to detect local and/or nodal regrowth. Computerized tomography (CT) scan of the chest, abdomen and pelvis should be done every 6 months during the first 3 years to detect distant metastases. The risk of both local and distant metastases is low after 3 years. Therefore, we do not recommended routine radiological examinations unless there is suspicion of a persistent tumor or development of distant metastases[7]. We advocate regular follow-up of the patients in the center where the treatment was delivered initially, by the same observer (if possible) or by a dedicated clinician following a “watch and wait” program. Patient follow-up also can be performed at the referring center, alternating with the CXB treatment center, by a limited number of clinicians who are experienced in the watchful waiting protocol. The radiological examinations should be done under a strict rectal protocol and reported by a radiologist familiar with the “watch and wait” clinical strategy, because interpretation of the images can sometimes be challenging. If necessary, these images should be referred to an experienced radiologist for review. Likewise, endoscopic examination should only be done by experienced clinicians familiar with the “watch and wait” follow-up process for these cancers. It is important not to biopsy normal mucosa or non-cancerous radiation-induced ulcers as the negative predictive value of a benign rectal biopsy is of very limited value. Moreover, complications such as perforations, delays in wound healing, protracted bleeding, or persistent pain can occur if the tumor is very low in the rectum[9]. In addition, fibrosis following a biopsy can make the interpretation of the subsequent radiological images more difficult. If there is uncertainty regarding abnormalities, either on endoscopy or in the interpretation of radiological images, the best approach is to refer the patient back to an appropriate cancer center for further assessment. In uncertain cases, it is best to repeat the investigations sooner (within 6-8 weeks) to assess any changes and refer the patient back to the cancer center for an expert opinion. If there is local regrowth of the tumor, the appearance will change at that site, but the changes usually are subtle and progress slowly. Examination under anesthesia for a targeted deep biopsy may be necessary to identify local regrowth, but this is not mandatory, as most regrowth are embedded deep within the muscles (muscularis propria) and it is not always possible to get the histological evidence of local regrowth unless the whole area is removed surgically.
CXB for local persistence of tumors after EBCRT
The watch and wait protocol with deferred surgery can be offered to patients who achieve cCR following EBCRT or EBRT. However, the majority of patients (74%) have residual tumor reported following EBCRT or EBRT[6] and the standard of care is to offer these patients surgery. However, if the patient is a not suitable surgical candidate or still refuses surgery, CXB can be offered as a booster therapy. There is published evidence that some of these patients can achieve cCR following CXB boost for their residual tumor[4,7]. Patients can then be follow up by the ‘watch and wait’ strategy and avoid immediate surgery.
Residual tumors after CXB and EBCRT
If there is residual tumor following EBRT and CXB boost, surgery can then be offered. For small residual tumors, trans-anal endoscopic microscopic surgery (TEMS) can be offered, because a proportion of residual mucosal abnormalities turn out to be benign adenomas that are difficult to differentiate from residual adenocarcinomas[10]. For gross residual tumors, salvage total mesorectal excision (TME) surgery should be offered[11]. However, the patient may not be medically fit for TME surgery or may refuse it. However, in our experience, at this stage most patients will agree for surgery, as they have tried the alternative non-surgical route and accept that this has failed. It is important to stress to the patient during the informed consent process that not all rectal cancers respond to CXB boosts after their EBRT, and that they may need to undergo salvage surgery if there is persistent residual tumor or a local growth at a later date[4,7].
Surgical salvage for local regrowth after cCR following EBCRT or EBRT and CXB
Local regrowth of a rectal cancer after achieving cCR following EBCRT or EBRT and CXB boost can be successfully treated if the patient is fit and agrees to surgery. Unfortunately, not all patients with local regrowth are fit and willing to undergo surgery[8]. Local regrowth following EBCRT or EBRT and CXB reportedly occurs in 11%-12% of cases[4,6,12,13], a rate that is much lower than the 25%-38% local regrowth that has been reported following EBCRT or EBRT alone[3,5].
Discussion
Most colorectal cancer treatment protocols and guidelines do not include radiotherapy for early rectal neoplasms[1,2]. Most colorectal MDT recommendations do not advocate non- surgical treatment even for early rectal cancers detected by screening. The dilemmas arise when a patient refuse the MDT recommendations. The UK National Institute for Health and Clinical Excellence (NICE) guidelines state that patients can refuse medical interventions to the extent of electing to undergo no treatment[14]. Most clinicians will only consider alternative treatment options if there is no evidence from a randomized trial. It is not always possible to do a randomized trial when two treatment strategies are not in equipoise with entirely different outcomes. In the absence of data from “hard to do” randomized trials, we need to consider how best to gather evidence to support the watch and wait approach. Most patients prefer not to have a stoma if there is a choice. The management of rectal cancer is becoming more complex and all cases should be discussed at the colorectal MDT before any treatment is offered. All treatment options that are available should be explained to the patients and their caregivers so that genuine “shared decision making” occurs before consent for treatment is obtained[15]. Sufficient time should be given to the patient prior to making that decision. Clinicians should be aware that some patients cannot handle too much information, and provision of needed but not excessive information to these patients must be considered. However, enough information should be given so as to allow the patient to make choices that take into account their values, which can be quite different from established medical views. In cases where uncertainties exist, the patients should be encouraged to participate in ongoing clinical trials so that meaningful data can be generated to help with decision-making in the future.
Following treatment, it is sometimes difficult to assess the clinical response, especially if the clinicians are not experienced in following a watch and wait strategy. Newer cancer centers that are starting to adopt these non-surgical treatment plans should work closely with, and take advice from, more experienced clinicians at other cancer hospitals. Not all patients with mucosal abnormalities have residual tumors[8] and clinicians should be aware that not all abnormalities on MRI represent a residual tumor. There are many uncertainties and clinicians should be encouraged to work closely with oncologists at nearby cancer centers who have more experience, so as to avoid performing unnecessary salvage surgeries, which can be devastating for the patient when there is no residual cancer. Litigation could follow, and so the possibility of this scenario should be clearly explained to the patient[16]. In cases where there is clinical uncertainty, it is better to wait a little longer to clarify the situation, to determine whether or not there is regrowth of any residual tumor, as the regrowth does not progress as quickly as one would expect.
Conclusion
The management of rectal cancer is becoming complex, even for early-stage tumors, and all cases should be presented and discussed in an early rectal MDT. Patients have a right to refuse the MDT recommendations, and alternative treatment options should be presented and explained to the patients and their caregivers. Patients should be made aware of any uncertainties about the possible treatments, including lack of data from relevant randomized trials that might guide rational evidence-based decisions. The rectal cancer patients should be encouraged to enter into ongoing clinical trials and ongoing trials such as the Organ Preservation for Early Rectal Adenocarcinoma trial (OPERA)[17] which may provide some useful data for decision making in the future.
Declarations
Author’s contributionsBoth authors contributed equally to all.
Availability of data and materialsThe data were strictly obtained from medical records according to the privacy policy and ethics code of our institute. All materials data are from Clatterbridge data base.
Financial support and sponsorshipNone.
Conflicts of interestGerard JP is the medical advisor for Ariane company. Myint AS has no conflict of interest.
Ethical approval and consent to participateEthic approval (01-02/26). Consent was obtained in this study.
Consent for publicationNot applicable.
Copyright© The Author(s) 2018.
REFERENCES
1. Glynne-Jones R, Wyrwicz L, Tiret E, Brown G, Rödel C, Cervantes A, Arnold D; ESMO Guidelines Committee. Rectal cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol 2017;28:iv22-40.
2. Moran B, Karandikar S, Geh I. Association of coloproctology of great Britain & Ireland (ACPGBI): guidelines for the management of cancer of the colon, rectum and anus (2017) - introduction. Colorectal Dis 2017;19:6-8.
3. Renehan AG, Malcomson L, Emsley R, Gollins S, Maw A, Myint AS, Rooney PS, Susnerwala S, Blower A, Saunders MP, Wilson MS, Scott N, O'Dwyer ST. Watch-and-wait approach versus surgical resection after chemoradiotherapy for patients with rectal cancer (the OnCoRe project): a propensity-score matched cohort analysis. Lancet Oncol 2016;17:174-83.
4. Sun Myint A, Smith FM, Gollins SW, Wong H, Rao C, Whitmarsh K, Sripadam R, Rooney P, Hershman MJ, Fekete Z, Perkins K, Pritchard DM. Dose escalation using contact X-ray brachytherapy (Papillon) for rectal cancer: does it improve the chance of organ preservation? B J Radiol 2017;90:20170175.
5. Habr-Gama A, Gama-Rodrigues J, São Julião GP, Proscurshim I, Sabbagh C, Lynn PB, Perez RO. Local recurrence after complete clinical response and watch and wait in rectal cancer after neoadjuvant chemoradiation: impact of salvage therapy on local disease control. Int J Radiat Oncol Biol Phys 2014;88:822-8.
6. Duldulao MP, Lee W, Streja L, Chu P, Li W, Chen Z, Kim J, Garcia-Aguilar J. Distribution of residual cancer cells in the bowel wall after neoadjuvant chemoradiation in patients with rectal cancer. Dis Colon Rectum 2013;56:142-9.
7. Sun Myint A, Smith FM, Gollins S, Wong H, Rao C, Whitmarsh K, Sripadam R, Rooney P, Hershman M, Pritchard DM. Dose escalation using contact X-ray brachytherapy after external beam radiotherapy as nonsurgical treatment option for rectal cancer: outcomes from a single-center experience. Int J Radiat Oncol Biol Phys 2018;100:565-73.
8. van der Valk MJM, Hilling DE, Bastiaannet E, Meershoek-Klein Kranenbarg E, Beets GL, Figueiredo NL, Habr-Gama A, Perez RO, Renehan AG, van de Velde CJH; IWWD Consortium. Long-term outcomes of clinical complete responders after neoadjuvant treatment for rectal cancer in the International Watch & Wait Database (IWWD): an international multicentre registry study. Lancet 2018;391:2537-45.
9. NICE. Low energy contact X-ray brachytherapy (the Papillon technique) for early-stage rectal cancer. Available from: http://www.contactpapillon.com/wp-content/uploads/2016/04/NICE-Papillon-guidance-Sept-2015.pdf. [Last accessed on 17 Sep 2018].
10. Smith FM, Chang KH, Sheahan K, Hyland J, O'Connell PR, Winter DC. The surgical significance of residual mucosal abnormalities in rectal cancer following neoadjuvant chemoradiotherapy. Br J Surg 2012;99:993-1001.
11. Hershman MJ, Sun Myint A. Salvage surgery after inadequate combined local treatment for early rectal cancer. Clin Oncol (R Coll Radiol) 2007;19:720-3.
12. Dhadda AS, Martin A, Killeen S, Hunter IA. Organ preservation using contact radiotherapy for early rectal cancer: outcomes of patients treated at a single centre in the UK. Clin Oncol (R Coll Radiol) 2017;29:198-204.
13. Frin AC, Evesque L, Gal J, Benezery K, François E, Gugenheim J, Benizri E, Château Y, Marcié S, Doyen J, Gérard JP. Organ or sphincter preservation for rectal cancer. The role of contact X-ray brachytherapy in a monocentric series of 112 patients. Eur J Cancer 2017;72:124-36.
14. NICE. Colorectal cancer: diagnosis and management. Available from: https://www.nice.org.uk/guidance/cg131.[Last accessed on 17 Sep 2018].
15. General Medical Council. Ethical guidance for doctors. Available from: https://www.gmc-uk.org/ethical-guidance/ethical-guidance-for-doctors.[Last accessed on 17 Sep 2018].
16. BMJ Publishing Group Ltd. When surgeons unwittingly don't obtain informed consent. Available from: https://blogs.bmj.com/bmj/2017/10/06/fraser-smith-and-david-locke-when-surgeons-unwittingly-dont-obtain-informed-consent/.[Last accessed on 17 Sep 2018].
17. U.S. National Library of Medicine. European phase III study comparing, in association with neoadjuvant chemoradiotherapy, a radiation dose escalation using 2 different approaches: external beam radiation therapy versus endocavitary radiation therapy with contact X-ray brachytherapy 50 kV for patients with rectal adenocarcinoma cT2-T3 a,b < 5cm in diameter in distal and middle rectum. Organ preservation for early rectal adenocarcinoma. Available from:https://www.clinicaltrials.gov.[Last accessed on 17 Sep 2018].
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Myint AS, Gerard JP. Minimally invasive contact X-ray brachytherapy as an alternative option in patients with rectal cancer not suitable for bespoke surgical resection. Mini-invasive Surg 2018;2:34. http://dx.doi.org/10.20517/2574-1225.2018.52
AMA Style
Myint AS, Gerard JP. Minimally invasive contact X-ray brachytherapy as an alternative option in patients with rectal cancer not suitable for bespoke surgical resection. Mini-invasive Surgery. 2018; 2: 34. http://dx.doi.org/10.20517/2574-1225.2018.52
Chicago/Turabian Style
Myint, Arthur Sun, Jean Pierre Gerard. 2018. "Minimally invasive contact X-ray brachytherapy as an alternative option in patients with rectal cancer not suitable for bespoke surgical resection" Mini-invasive Surgery. 2: 34. http://dx.doi.org/10.20517/2574-1225.2018.52
ACS Style
Myint, AS.; Gerard JP. Minimally invasive contact X-ray brachytherapy as an alternative option in patients with rectal cancer not suitable for bespoke surgical resection. Mini-invasive. Surg. 2018, 2, 34. http://dx.doi.org/10.20517/2574-1225.2018.52
About This Article
Special Issue
Copyright
Data & Comments
Data
 Cite This Article 5 clicks
Cite This Article 5 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.