The role of splenic flexure mobilization in laparoscopic rectal surgery for rectal cancer
Abstract
Laparoscopic surgery for the treatment of colorectal cancer has gained an enormous advantage as compared to the traditional approach in terms of patient benefits. Although it has gained popularity among surgeons, there are still some contentious issues especially in laparoscopic rectal surgery. Splenic flexure mobilization is a crucial aspect of the procedure with complex technical details thereby establishing a learning curve that cannot be easily overcome. A team of colorectal surgeons at China Medical University Hospital adopted a standardized approach to laparoscopic rectal surgery particularly simplifying the steps involved in mobilizing the splenic flexure which is deemed as one of the difficult steps in the surgery.
Keywords
Introduction
One of the most commonly diagnosed malignancies in the world is colorectal cancer (CRC). It currently ranks third based on the GLOBOCAN (Global Cancer Incidence, Mortality and Prevalence) index and is also the fourth leading cause of cancer-related deaths. The cancer burden will be increasing by 60% to more than 2.2 million new cases and resulting in 1.1 million deaths by 2030[1]. Based on the National Cancer Registry of Taiwan, CRC is the second most common invasive neoplastic disease with a total of 15,764 cases in 2014 and a crude incidence rate of 67.27%[2]. The evolution of laparoscopic-assisted compared to open approach colorectal surgery for diverticular disease and cancer was first introduced in the early 1990s and was aimed to offer the benefit of less trauma, without compromising functional and oncological outcomes[3]. Laparoscopic surgery has gained increasing interest for the treatment of CRC. Laparoscopic approach for colon and rectal cancer was associated with less morbidity, enhanced recovery, and at least equivalent oncological outcomes[3-6]. In China Medical University Hospital (CMUH), a tertiary medical center in Taichung City, a team of colorectal surgeons performs a high volume of cases for laparoscopic rectal surgeries.
Left-sided CRC comprises two thirds of all colorectal malignancies. The standard surgical treatment is a complete oncologic resection with a primary anastomosis[7]. There were initial concerns about the potential risk of tumor cell dissemination during laparoscopy but this has not been validated. The emergence of laparoscopic colorectal surgery has not been fully embraced by most surgeons mostly because of the increased technical laparoscopic skill requirements[4]. This paper aims to discuss the importance of splenic flexure mobilization (SFM) and its technical details during laparoscopic rectal surgery.
SFM
SFM is one of the essential, challenging and technically demanding step during laparoscopic rectal cancer surgery. The use of SFM for CRC surgery remains a contentious issue[5,6,8], but safe dissection of the splenic flexure to fully mobilize the descending colon is mandatory not only for oncologic resection but also for safe anastomosis[4,9-11]. The definition of SFM is different among several studies with some describing the technique as either complete or partial mobilization[3,5,6,12]. However, it is relevant to know the procedural aspect of SFM consisting of the division of the splenocolic, phrenicocolic, gastro colic and pancreaticomesocolic ligaments. It is crucial to differentiate a partial splenic flexure from the complete SFM. In partial SFM, it is limited only to the division of splenocolic and phrenicocolic ligaments while a complete SFM includes not only the division of splenocolic and phrenicocolic ligaments (partial mobilization) but the division of gastro-colic and pancreaticomesocolic attachments. This can be technically accomplished either through a lateral-to-medial or a medial-to-lateral approach[3,4,6,9,10]. A variety of approaches for SFM have been used by surgeons to simplify the technique. A median to lateral approach for the complete mobilization of the splenic flexure was commonly described in various studies[9,10]. This approach is similar to the study of Marsden et al.[13], in which many surgeons favor routine mobilization of the flexure at an early stage in the operation, particularly for low rectal cancers. It is often considered helpful to carry out this step along with division of the inferior mesenteric artery (IMA), the inferior mesenteric vein (IMV) and the colon before beginning the pelvic dissection. This approach allows the divided colon and small bowel to be packed away giving good access to the pelvis for the rectal dissection[4,9,10]. This highlights the importance of a complete SFM prior to the pelvic dissection of any rectal surgery.
SFM is a crucial part in all left-sided colorectal surgeries particularly laparoscopic anterior and low anterior resections[4,9,10]. SFM is performed in order to achieve adequate oncological resection, create a tension-free anastomosis with a good blood supply, and perform a pouch reconstruction if necessary[3,4,14]. It allows to achieve a straight segment of supple and well vascularized segment of the descending colon that can be easily anastomosed to the remnant rectum down in the pelvis in which some surgeons favor creating a recreational pouch to decrease frequency of bowel movement[5]. In a cadaveric study done by Thum-umnuaysuk et al.[7], a greater length of colon at 17.98 ± 6.80 cm was achieved and it reached statistical significance when high ligation of IMA and IMV coupled with SFM was done. In a separate cadaveric study by Araujo et al.[11], it was shown that an additional 10 to 28 cm segment of the descending colon can be gained if SFM was carried out with or without distal transverse colon mobilization. Kye et al.[15] cited that as much as 30 centimeters of colon redundancy will be reached if high IMV ligation was performed as compared to a low IMV ligation which gains 5 centimeters less. The results in the paper of Kye et al.[15] had comparable results to the previous cadaveric studies which considered SFM as vital in every laparoscopic rectal surgery to come up with a lengthy colon needed to have a tension-free anastomosis. Elongation of the colon is essential in creating a tension-free anastomosis which involves adequate mobilization of the bowel ends particularly on the colonic side[16]. The vascular supply of the proximal and distal margins after resection becomes an integral part of the process[8-20]. In the process of performing a complete SFM, it is noteworthy that only high ligation of both the IMV and IMA as well as division of the involved attachments in the splenic flexure will result in a lengthy colon that would provide for a tension-free anastomosis. Toh et al.[17], highlighted the presence of an important vessel known as the Arc of Riolan that provides collateral mesenteric circulation in 10% of individuals and naturally found anterior to the IMV on the inferior border of the pancreas. It is important that this vessel should be preserved during high ligation of the IMV and splenic flexure takedown to ensure collateral supply from the SMA through the connection between the middle colic and left colic artery[17]. Performing a technically sound and complete SMF can have an impact on the patient’s postoperative recovery although the success of the surgery is reliant on several compounding factors.
Several of these studies mentioned a variety of possible risk factors owing to the technical difficulty of such a procedure. The presence of comorbidities such as hypertension and diabetes mellitus, increased BMI, increased American Society of Anesthesiologist score, previous neoadjuvant chemo radiotherapy have been linked to the technical difficulty of performing SFM due to the risks that accompany it. However, the results did not reach statistical significance leading to the conclusion that the benefits of the step in all laparoscopic rectal surgeries far outweighs the risk[4-6,8-10,12,14-16]. One common intraoperative complication cited in various studies is causing tears to the spleen whether it is complete or partial avulsion will be immaterial since both can cause significant bleeding[4-6,8-10,12]. This particularly happens during the process of dividing the phrenicocolic and especially the splenocolic ligaments. There were mentions of inadvertent serosal injuries to the small bowel but they were insignificant as far as the overall result of those studies were concerned[5,12,18]. The presence of anastomotic leaks after laparoscopic rectal surgery in which SFM was performed were cited in some of the published papers[8,12,15,16,18]. It was attributed in some cases to the presence of tension and inadequate vascularity on the involved segments but no direct links were established between SFM and anastomotic leaks since it is considered to be multifactorial.
The team of colorectal surgeons in CMUH adopted the method of doing a mandatory SFM. Incorporating SFM in all laparoscopic rectal surgeries will enable the team to overcome the learning curve involved in this very technical procedure.
Surgical method
The following part details the precise description of how a complete SFM is done in our institution. The approach utilized is a combined medial to lateral and lateral to medial dissection with emphasis on the ease of performing the steps during laparoscopic surgery.
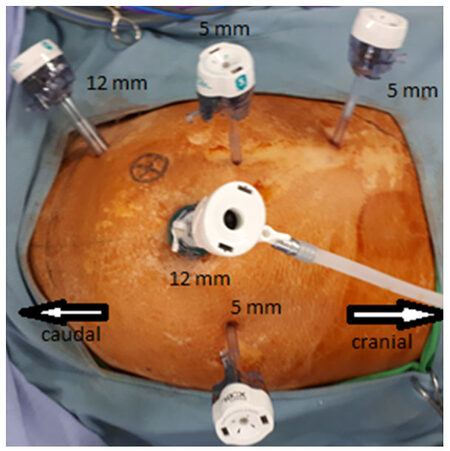
The patient is placed in a Trendelenburg and a semi-right lateral decubitus position. The patient is prepped in a conventional manner and a conventional 5-trocar placement is instituted. A 12-mm trocar is inserted at the umbilicus employing the Hasson’s technique (entering the abdomen under direct visualization) as well as at the right lower quadrant of the abdomen. Additional 5 mm trocars are placed each at the right and left paraumbilical area (5-6 cm from the umbilicus) as well as on the right upper quadrant of the abdomen along the mid-clavicular line [Figure 1].
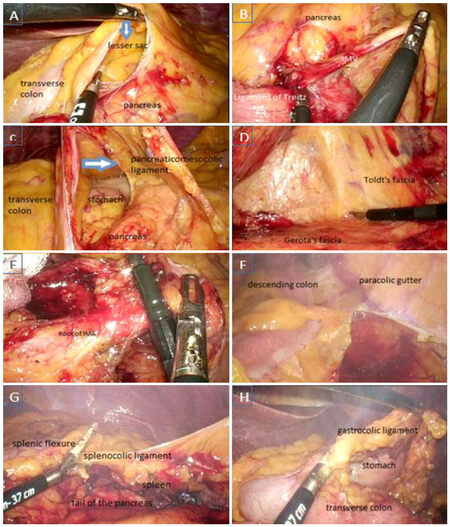
After a thorough inspection of the abdominal cavity, the small bowel is carefully placed to the right side of the abdomen using atraumatic bowel graspers and exposing the ligament of Treitz where the root of the IMV is also located. The inferior border of the pancreas is likewise identified and the mesentery is carefully grasped and sharp dissection is initiated using monopolar electro cautery to enter the lesser sac. This is followed by incising the paraaortic peritoneum and the IMV is ligated at its root and divided using Ligasure. The retroperitoneal dissection is carried out over the Gerota’s fascia by carefully separating the Toldt’s fascia aided by sharp dissection until the mesentery of the descending colon can be lifted up to form a tent. The pancreaticomesocolic attachments along the tail of the pancreas is carefully divided using Ligasure to render visible the splenic hilum [Figure 2A-D].
Figure 2. Splenic flexure mobilization. A: Entering the lesser sac by dividing the mesentery at the inferior border of the pancreas; B: high ligation of the inferior mesenteric vein proximal to the ligament of Treitz and on the inferior border of the pancreas; C: division of the pancreaticomesocolic ligament; D: retroperitoneal dissection and separation of the Toldt’s fascia; E: ligation of the inferior mesenteric artery at its root; F: lateral dissection involving division of the left paracolic gutter; G: division of the splenocolic ligament; H: completion of splenic flexure mobilization by division of the gastrocolic ligaments
The IMA is identified and isolated at its take off from the abdominal aorta. The IMA is ligated at around 1-1.5 cm distal to the aorta and carefully divided using Ligasure. Posterior dissection is continued caudally while preserving all identified retroperitoneal structures along the dissection until the presacral space is reached. Sharp dissection is carried out laterally until reaching the left paracolic gutter where the parietal peritoneum commences. The sigmoid and descending colon is now mobilized by dividing the parietal peritoneum from the pelvis until the hilum of the spleen is visible. SFM is completed by detaching the omentum from the transverse colon and dividing the splenocolic and gastro colic ligaments [Figure 2E-H].
In the pelvic phase of all laparoscopic rectal surgeries, an additional 5-mm trocar can be inserted in the left lower quadrant of the abdomen that can be used by the assistant surgeon during this phase of the dissection. This is particularly done especially in low lying rectal tumors where a laparoscopic low anterior resection or transanal total mesorectal excision will be performed.
Conclusion
In our perspective, SFM is an integral step in performing laparoscopic surgery for rectal cancer. This will enable the surgeon to achieve a tension-free anastomosis from an adequate redundant colon and have good vascularity on both the proximal and distal ends of the segment. There is a learning curve involved in such procedure and it can easily be overcome in high volume centers such as our institution where the steps can be readily performed.
Declarations
Authors’ contributionsPerformed the operations: Ke TW, Geniales CR, Chen WTL
Participated in the data and patients collection, wrote the manuscript: Ke TW, Geniales CR
Supervised this study: Chen WTL
Availability of data and materialsNot applicable.
Financial support and sponsorshipNone.
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Author(s) 2018.
REFERENCES
1. Arnold M, Sierra MS, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global patterns and trends in colorectal cancer incidence and mortality. Gut 2017;66:683-91.
2. Taiwan Cancer Registry. Available from: http://tcr.cph.ntu.edu.tw/main.php?Page=N1. [Last accessed on 15 Sep 2018].
3. Vecchio R, Marchese S, Intagliata E. Laparoscopic colorectal surgery for cancer: what is the role of complete mesocolic excision and splenic flexure mobilization? Indian J Surg 2017;79:338-43.
4. Gouvas N, Gogos-Pappas G, Tsimogiannis K, Agalianos C, Tsimoyiannis E, Dervenis C, Xynos E. Impact of splenic flexure mobilization on short-term outcomes after laparoscopic left colectomy for colorectal cancer. Surg Laparosc Endosc Percutan Tech 2014;24:470-4.
5. Kim MK, Lee IK, Kang WK, Cho HM, Kye BH, Jalloun HE, Kim JG. Long-term oncologic outcomes of laparoscopic surgery for splenic flexure colon cancer are comparable to conventional open surgery. Ann Surg Treat Res 2017;93:35-42.
6. Gezen C, Altuntas YE, Kement M, Vural S, Civil O, Okkabaz N, Aksakal N, Oncel M. Complete versus partial mobilization of splenic flexure during laparoscopic low anterior resection for rectal tumors: a comparative study. J Laparoendosc Adv Surg Tech A 2012;22:392-6.
7. Thum-umnuaysuk S, Boonyapibal A, Geng YY, Pattana-Arun J. Lengthening of the colon for low rectal anastomosis in a cadaveric study: how much can we gain? Tech Coloproctol 2013;17:377-81.
8. Schlussel AT, Wiseman JT, Kelly JF, Davids JS, Maykel JA, Sturrock PR, Sweeney WB, Alavi K. Location is everything: the role of splenic flexure mobilization during colon resection for diverticulitis. Int J Surg 2017;40:124-29.
9. Kim HJ, Kim CH, Lim SW, Huh JW, Kim YJ, Kim HR. An extended medial to lateral approach to mobilize the splenic flexure during laparoscopic low anterior resection. Colorectal Dis 2013;15:e93-8.
10. Benseler V, Hornung M, Iesalnieks I, von Breitenbuch P, Glockzin G, Schlitt HJ, Agha A. Different approaches for complete mobilization of the splenic flexure during laparoscopic rectal cancer resection. Int J Colorectal Dis 2012;27:1521-9.
11. Araujo SE, Seid VE, Kim NJ, Bertoncini AB, Nahas SC, Cecconello I. Assessing the extent of colon lengthening due to splenic flexure mobilization techniques: a cadaver study. Arq Gastroenterol 2012;49:219-22.
12. Zhou T, Zhang G, Tian H, Liu Z, Xia S. Laparoscopic rectal resection versus open rectal resection with minilaparotomy for invasive rectal cancer. J Gastrointest Oncol 2014;5:36-45.
13. Marsden MR, Conti JA, Zeidan S, Flashman KG, Khan JS, O'Leary DP, Parvaiz A. The selective use of splenic flexure mobilization is safe in both laparoscopic and open anterior resections. Colorectal Dis 2012;14:1255-61.
14. Matsumura N, Tokumura H, Saijo F, Katayose Y. Strategy of laparoscopic surgery for colon cancer of the splenic flexure: a novel approach. Surg Endosc 2018;32:2559.
15. Kye BH, Kim HJ, Kim HS, Kim JG, Cho HM. How much colonic redundancy could be obtained by splenic flexure mobilization in laparoscopic anterior or low anterior resection? Int J Med Sci 2014;11:857-62.
16. Reddy SH, Gupta V, Yadav TD, Singh G, Sahni D. Lengthening of left colon after rectal resection: what all is adequate? A prospective cohort study. Int J Surg 2016;31:27-32.
17. Toh JWT, Matthews R, Kim SH. Arc of riolan-preserving splenic flexure takedown during anterior resection: potentially critical to prevent acute anastomotic ischemia. Dis Colon Rectum 2018;61:411-4.
18. Okuda J, Yamamoto M, Tanaka K, Masubuchi S, Uchiyama K. Laparoscopic resection of transverse colon cancer at splenic flexure: technical aspects and results. Updates Surg 2016;68:71-5.
19. Ludwig KA, Kosinski L. Is splenic flexure mobilization necessary in laparoscopic anterior resection? Another view. Dis Colon Rectum 2012;55:1198-200.
20. Murpurgo E, Contardo T, Termini B, Orsini C, Tosato SM, Annibale AD. Laparoscopic takedown of the splenic flexure. Available from: http://cine-med.com/index.php?id=acs-2721. [Last accessed on 15 Sep 2018].
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Ke TW, Geniales CR, Chen WL. The role of splenic flexure mobilization in laparoscopic rectal surgery for rectal cancer. Mini-invasive Surg 2018;2:35. http://dx.doi.org/10.20517/2574-1225.2018.46
AMA Style
Ke TW, Geniales CR, Chen WL. The role of splenic flexure mobilization in laparoscopic rectal surgery for rectal cancer. Mini-invasive Surgery. 2018; 2: 35. http://dx.doi.org/10.20517/2574-1225.2018.46
Chicago/Turabian Style
Ke, Tao-Wei, Christian Ross Geniales, William Tzu-Liang Chen. 2018. "The role of splenic flexure mobilization in laparoscopic rectal surgery for rectal cancer" Mini-invasive Surgery. 2: 35. http://dx.doi.org/10.20517/2574-1225.2018.46
ACS Style
Ke, T.W.; Geniales CR.; Chen W.L. The role of splenic flexure mobilization in laparoscopic rectal surgery for rectal cancer. Mini-invasive. Surg. 2018, 2, 35. http://dx.doi.org/10.20517/2574-1225.2018.46
About This Article
Special Issue
Copyright
Data & Comments
Data
 Cite This Article 17 clicks
Cite This Article 17 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.