Laparoscopic vs. open distal gastrectomy for advanced gastric cancer in elderly patients: a retrospective study
Abstract
Aim: It is unclear whether elderly patients with advanced gastric cancer can benefit from laparoscopic gastrectomy. This study aimed to compare the surgical and early postoperative outcomes of laparoscopic distal gastrectomy with those of open distal gastrectomy for advanced gastric cancer in elderly patients aged 75 years or older.
Methods: We retrospectively examined all elderly patients who underwent laparoscopic distal gastrectomy or open distal gastrectomy from October 2010 to October 2017 using prospectively collected data. Operative results, hospital courses, and survival rates were compared between the two groups.
Results: Distal gastrectomy was performed in 60 patients, laparoscopically in 20 and through open surgery in 40. The laparoscopic group had significantly lesser intraoperative blood loss (100 mL vs. 300 mL; P < 0.001) and shorter mean postoperative hospital stays (12 days vs. 23 days; P < 0.001). The overall 3-year survival rate was 50.1% in the laparoscopic group and 41.7% in the open group (P = 0.531).
Conclusion: Laparoscopic distal gastrectomy led to a faster return to a full diet and a shorter postoperative hospital stay in our study, and it was well tolerated by elderly patients with advanced gastric cancer.
Keywords
Introduction
In Japan, the 2017 Annual Health, Labour and Welfare Report defined an “elderly” or older person as an individual aged 75 years or older. The average age of the population and the number of elderly patients have been increasing worldwide, particularly in developing countries[1]. Elderly patients usually have various comorbidities such as cardiovascular diseases[2,3] and decreased respiratory function, making them unsuitable for surgery. Age exceeding 70 years is an independent predictor of increased postoperative complications, in-hospital mortality, and longer hospital stays[4-6].
The advantages of laparoscopic surgery demonstrated by several reports include a decreased morbidity rate, decreased pain, and faster recovery[7-9]. It is important to determine whether elderly patients with advanced gastric cancer can benefit from laparoscopic surgery. However, few studies on laparoscopic distal gastrectomy (LDG) in elderly patients, especially those with advanced gastric cancer, have been reported. Therefore, it is necessary to study the safety, efficacy, and outcomes of LDG in elderly patients with advanced gastric cancer. Although several studies have reported the safety and benefits of laparoscopic surgery for gastric cancer in the elderly, most authors preferred to compare the findings in the elderly with those in younger patients[10,11]. These researchers could not illustrate that the laparoscopic surgery for gastric cancer in elderly patients were more effective and safe than traditional open surgery. Therefore, the present study aimed to compare the surgical and early postoperative outcomes of LDG with those of open distal gastrectomy (ODG) for advanced gastric cancer in patients age 75 years or older.
Methods
We identified 60 patients aged 75 years or older[12] who underwent LDG or ODG for advanced primary gastric cancer at the Department of Surgery of Bell Land General Hospital from October 2010 to October 2017. The patients were retrospectively selected from a prospectively collected database and divided into two groups based on the operative approach: the LDG group and the ODG control group.
Exclusion criteria included urgent or emergent procedures and operations other than distal gastrectomy, such as total or proximal gastrectomy. Preoperative diagnosis was made by upper endoscopy with tumor biopsy, and clinical staging was performed with abdominopelvic computed tomography. Tumors were staged according to the Japanese Classification of Gastric Carcinoma[12].
In this study, laparoscopic surgery was performed by three surgeons who were proven experts in their field as defined by the Japan Society for Endoscopic Surgery. Two of the three surgeons are experts in laparoscopic gastrectomy. The indications for LDG and ODG were the same: clinically diagnosed gastric cancer without distant metastasis and lymph node involvement in the extraperigastric area. The patients were fully informed of their diagnoses and briefed on whether they would undergo LDG or ODG. The choice of LDG or ODG was decided upon by the patient and the attending surgeon after discussing both approaches. Written informed consent was obtained from all patients before the operation.
Data on several factors, including preoperative patient baseline parameters, perioperative variables, postoperative outcomes, and pathologic results, were collected for analysis. The preoperative parameters analyzed were age, sex, body mass index (BMI), American Society of Anesthesiology (ASA) classification, previous abdominal surgery, and comorbidities, which were assessed using the Charlson Comorbidity Index (CCI)[13]. Perioperative variables analyzed included the mode of anastomosis, length of the operation, estimated blood loss, time to first oral diet, and duration of postoperative hospital stay. Postoperative outcomes included postoperative complications, 30-day mortality, and recurrences. Postoperative complications, such as anastomotic leakage and pancreatic fistula, were classified based on the Clavien-Dindo classification[14]. In addition, postoperative complications such as pneumonia and delirium, which are especially critical in elderly patients, were included in the analysis.
This study was approved by the institutional review board of the Bell Land General Hospital. The study protocol conformed to the principles set in the Declaration of Helsinki and its later amendments.
All statistical analysis were performed with EZR (Saitama Medical Center, Jichi Medical University; http://www.jichi.ac.jp/saitama-sct/SaitamaHP.files/statmedEN.html; Kand, 2012), which is a graphical user interface for R (version 2.13.0; The R Foundation for Statistical Computing, Vienna, Austria)[15].
The characteristics of both groups were compared using the chi-squared and Mann-Whitney U tests. The postoperative survival was analyzed using Kaplan-Meier survival curves and compared with the log-rank test. Significance was established at P < 0.05. As the number of patients older than 75 years are low, the sample size of the present study was small. Therefore, we used non-parametric statistical methods.
Results
Patient characteristics
Patient characteristics are shown in Table 1. There were 20 patients in the LDG group and 40 in the ODG group with mean ages of 81.0 ± 5.0 and 81.4 ± 3.9 years, respectively. No significant differences were observed in terms of sex, BMI, ASA classification, previous surgery, or CCI. Since decreased respiratory function is a major risk factor for postoperative pulmonary complications in the elderly, previous studies commonly assessed the predicted functional expiratory volume in 1 s, predicted vital capacity, or arterial oxygen saturation[16,17]. In this study, we used the Hugh-Jones classification to assess respiratory function[18], and no differences were observed between the two groups. The prevalence of comorbidity was high in both groups; only 25.0% of the patients in the LDG group (n = 5) and 15.0% of those in the ODG group (n = 6) had no preoperative comorbidity (CCI = 0).
Patient demographics of the LDG group and ODG group
| Variable | LDG group (n = 20) | ODG group (n = 40) | P value |
|---|---|---|---|
| Age (years) | 81.0 ± 5.0 | 81.4 ± 3.9 | 0.289 |
| Sex, F/M | 5/15 | 13/27 | 1 |
| BMI | 22.3 ± 3.4 | 21.4 ± 3.0 | 0.289 |
| ASA | 0.551 | ||
| 1 | 2 | 3 | |
| 2 | 15 | 25 | |
| 3 | 3 | 12 | |
| Hugh-Jones classification | 0.117 | ||
| 1 | 12 | 13 | |
| 2 | 7 | 16 | |
| 3 | 1 | 9 | |
| 4 | 0 | 2 | |
| Previous surgeries | 9 | 9 | 0.134 |
| CCI | 0.566 | ||
| 0 | 5 | 6 | |
| 1 | 7 | 10 | |
| 2 | 7 | 16 | |
| 3 | 1 | 6 | |
| 4 | 0 | 2 |
The surgical procedure and early surgical outcomes are summarized in Table 2. A Billroth I anastomosis was performed in 12 patients in the LDG group and in 18 in the ODG group, while a Roux-en-Y anastomosis was performed in 8 and 20 patients, respectively. The remaining patients in the ODG group underwent a Billroth II anastomosis. There were no conversions to open surgery in the LDG group. D2 lymph node dissection was performed in 15 patients in the LDG group and in 26 patients in the ODG group. There was a significant reduction in the estimated blood loss in the LDG group compared with that in the ODG group (P < 0.001). The time to first oral diet was significantly shorter in the LDG group than in the ODG group (P = 0.00185).
Surgical and oncological outcomes of the LDG group and ODG group
| LDG group (n = 20) | ODG group (n = 40) | P value | |
|---|---|---|---|
| Mode of anastomosis | 0.473 | ||
| Billroth I | 12 | 18 | |
| Billroth II | 0 | 2 | |
| Roux-en-Y | 8 | 20 | |
| LN dissection | 0.77 | ||
| D1 | 2 | 9 | |
| D1+ | 3 | 5 | |
| D2 | 15 | 26 | |
| Operative time (min) | 328 ± 57 | 243 ± 57 | < 0.001 |
| EBL (mL, range) | 100 (0-475) | 300 (60-2,464) | < 0.001 |
| Time to first oral diet (days) | 4.9 ± 2.4 | 8.3 ± 8.1 | 0.0185 |
| Postoperative stay (days, range) | 12 (8-39) | 23 (10-514) | < 0.001 |
| Postoperative metastasis | 3 (15%) | 13 (32.5%) | 0.0766 |
Postoperative hospital stay was significantly shorter in the LDG group than in the ODG group (P < 0.001). The clinical pathway was used after gastrectomy in the hospital.
Postoperative complications
Postoperative complications occurred in 8 patients (40.0%) in the LDG group and in 24 patients (60.0%) in the ODG group (P = 0.176). In terms of abdominal complications, only the rate of paralytic ileus, defined as bowel obstruction treated by a long nasogastric tube, was significantly lower in the LDG group (P = 0.040). No significant differences were found in the rates of other complications [Table 3]. Pneumonia occurred in 3 patients in the ODG group, but it did not occur in the LDG group (P = 0.544). Postoperative complications included all grades of Clavien-Dindo classification. A total of 4 patients (10.0%) in the ODG group died within 30 days due to postoperative complications: pneumonia in 2, anastomotic leakage in 1, and remnant stomach necrosis in 1. In the LDG group, only 1 patient died of enteritis due to methicillin-resistant Staphylococcus aureus on postoperative day 7.
Postoperative complications in the LDG group and ODG group
| LDG group (n = 20) | ODG group (n = 40) | P value | |
|---|---|---|---|
| Abdominal morbidity1 | |||
| Anastomotic leakage | 0 | 3 (7.5%) | 0.544 |
| Pancreatic fistula | 3 (15.0%) | 4 (10.0%) | 0.676 |
| Abdominal abcess | 1 (5.0%) | 3 (7.5%) | 1 |
| Anastomotic ulcer | 2 (10.0%) | 1 (2.5%) | 0.272 |
| Wound dehiscence | 0 | 1 (2.5%) | 1 |
| Paralytic ileus | 0 | 5 (12.5%) | 0.040 |
| Other morbidity1 | |||
| Wound infection | 0 | 6 (15.0%) | 0.083 |
| Pneumonia | 0 | 3 (7.5%) | 0.544 |
| Acute cardiac failure | 1 (5.0%) | 2 (5.0%) | 1 |
| Delirium | 1 (5.0%) | 3 (7.5%) | 1 |
| Overall | 8 (40.0%) | 24 (60.0%) | 0.176 |
| Mortality | 1 (5.0%) | 4 (10.0%) | 0.656 |
Pathological results
Table 4 describes the pathological findings in the 2 groups. No significant differences were noted in terms of depth of invasion, nodal catalog, and pathological stage. Stage 4 disease was observed in 1 patient in the LDG group and in 6 patients in the ODG group. Curative surgery was not performed in 7 patients: 1 patient who had liver metastasis (LDG group), 4 patients who had dissemination (ODG group), and 2 patients who had liver metastases (ODG group). No significant differences were found in terms of tumor diameter, the mean number of harvested lymph nodes, and the mean number of positive lymph node metastases between the two groups.
Pathological data of the LDG group and ODG group
| LDG group (n = 20) | ODG group (n = 40) | P value | |
|---|---|---|---|
| Depth of invasion | 0.156 | ||
| pT1 | 0 | 0 | |
| pT2 | 8 | 6 | |
| pT3 | 2 | 3 | |
| pT4a | 9 | 27 | |
| pT4b | 1 | 4 | |
| Nodal catalog | 0.215 | ||
| pN0 | 8 | 11 | |
| pN1 | 3 | 9 | |
| pN2 | 5 | 3 | |
| pN3a | 3 | 10 | |
| pN3b | 1 | 7 | |
| Stage | 0.62 | ||
| IA | 0 | 0 | |
| IB | 4 | 3 | |
| IIA | 1 | 4 | |
| IIB | 6 | 8 | |
| IIIA | 3 | 4 | |
| IIIB | 1 | 4 | |
| IIIC | 4 | 11 | |
| IV | 1 | 6 | |
| Diameter of tumor (cm) | 5.9 ± 2.2 | 6.1 ± 2.2 | 0.762 |
| Number of lymph node removed | 35.5 (14-65) | 33.5 (5-96) | 0.994 |
| Number of positive lymph node metastasis | 2 (0-12) | 2.5 (0-47) | 0.213 |
Prognosis
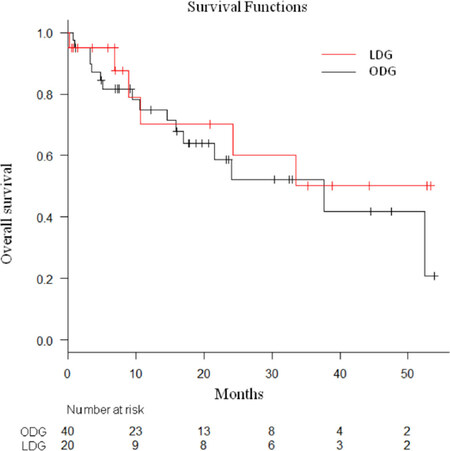
The LDG and ODG groups did not differ significantly in terms of the pathological stage according to the Japanese Classification of Gastric Carcinoma[12]. The median follow-up period in the LDG and ODG groups was 11.7 months (range, 0.3-54.8 months), and the 3-year overall survival was 50.1% and 41.7%, respectively (P = 0.531) [Figure 1].
Discussion
As life expectancy continues to increase, the number of elderly patients with malignancies and concomitant comorbidities has increased as well. The proportion of elderly patients among the total gastric cancer population is expected to increase gradually over the next few decades. As aging is associated with a gradual loss of reserve capacity[19], age exceeding 70 years is an independent risk factor for postoperative mortality, complications, and longer hospital stays after gastric cancer surgery[4,5].
According to the Japanese gastric cancer treatment guidelines, laparoscopic surgery is one of the most reliable treatments for early gastric cancer[20]. With the increase in the number of laparoscopic gastrectomies being performed, elderly patients might benefit from their less invasive nature. However, it is important to determine whether these advantages are applicable to elderly patients because the carbon dioxide pneumoperitoneum required for laparoscopy may be harmful and the patients’ frequent comorbidities and reduced physiological reserves indicate increased risk of postoperative morbidity and mortality[21,22]. Limited data exist on the efficacy of laparoscopic gastrectomy in elderly patients, especially those with advanced gastric cancer. We performed the current study to compare LDG in elderly patients with advanced gastric cancer to ODG and determine its feasibility and efficacy.
LDG for advanced gastric cancer was shown to be more effective than open surgery in elderly patients, resulting in reduced blood loss, faster first oral diet initiation, and shorter hospital stays. Conversely, the longer operative time may lead to a higher rate of morbidity, including delirium, pneumonia, and cardiac failure. Acute cardiac failure and pneumonia are the most common minor complications of distal gastrectomy for gastric cancer. Some reports have suggested that the adverse cardiopulmonary effects of pneumoperitoneum occur only when the intra-abdominal pressure is more than 15 mmHg, while low pressures do not affect cardiopulmonary output[23-25]. Therefore, laparoscopic surgery may be safe even in high-risk patients. However, the operative time was longer in the LDG group than in the ODG group, and longer operative times are associated with postoperative delirium and pneumonia[26]. Our results revealed no significant differences in postoperative morbidities, suggesting that high-risk patients, including the elderly may be able to tolerate the prolonged laparoscopic operative times. In accordance with previous reports, laparoscopic surgery led to a faster return to a full diet and a shorter postoperative hospital stay in our study[27].
In fact, a shorter abdominal incision leads to less pain and, subsequently, earlier ambulation. Early ambulation may prevent postoperative delirium as evidenced by the findings of Schweickert et al.[28], who found a shorter duration of intensive care unit-associated delirium in patients who received physical and occupational therapy. Since postoperative pain is one cause of delirium, less pain after surgery may help prevent postoperative delirium[29]. Therefore, it is more important to reduce pain after surgery than to shorten operative time, especially in elderly patients. In addition, early ambulation prevents pneumonia resulting from atelectasis. Based on these advantages, LDG may provide superior short-term outcomes in elderly patients with advanced gastric cancer.
A 2011 report stated that the average life expectancy at 80 years in Japan had increased by 8.39 years in men and 11.36 years in women. However, Endo et al.[2] reported that the overall survival of gastric cancer patients was longer in the operation group than in the best supportive care group. The oncologic results of LDG in elderly patients have not been determined. In a study of the overall gastric cancer patient population, Hu et al.[30] reported that the cumulative 3-year overall survival rate after laparoscopy-assisted gastrectomy for advanced gastric cancer was 75.3%; in our study, it was 50.1% and 41.7% in the LDG and ODG groups, respectively, which is lower than the overall rate. This may be because elderly patients have concurrent ailments that affect prognosis and survival. Indeed, in the present study, 49 patients (81.7%) had preoperative comorbid diseases. Concurrent disease was the cause of death in 2 patients in the LDG group and in 4 patients in the ODG group. Since elderly patients are expected to have a shorter residual life span, quality of life should also be considered when choosing a surgical procedure[31,32].
The elderly patients in the ODG group were more likely to require inpatient rehabilitation due to their diminished mobility and more persistent postoperative wound pain, which seriously hamper the recovery of activities of daily living[33]. Prolonged hospitalization after surgery can lead to worse quality of life in elderly patients. Several reports have demonstrated that laparoscopic surgery leads to a faster return to a full diet and a shorter postoperative hospital stay, as shown in our own study. These data indicate that LDG may be superior to conventional open surgery in terms of the length of the hospital stay, even for advanced gastric cancer.
Our overall rate of postoperative complications was higher than that in previous studies on the overall gastric cancer patient population. Hu et al.[30] reported a postoperative complication rate of 10.2% in 1,184 patients with advanced gastric cancer. The most common major complication in the LDG group was pancreatic fistula, which tends to reflect surgical inexperience and a learning curve[34]. Pancreatic fistulas occurred in 3 patients undergoing LDG in 2010, the year we started performing D2 lymphadenectomy. As experience accumulates, the incidence rate of major pancreatic fistulas may decrease. Although the mortality rate was not significantly different between the 2 groups, 4 deaths occurred in the ODG group, suggesting that radical gastrectomy may sometimes be excessively stressful for elderly patients, especially those with advanced gastric cancer. Surgery should be performed more meticulously and quickly than usual in elderly patients. The choice of surgical procedure in elderly patients with short life expectancies must guarantee disease control as well as acceptable short- and long-term survival and quality of life[35].
Our study had some limitations inherent to retrospective and non-randomized studies, where selection and observer biases with regard to the operative approach adopted are possible. Furthermore, our study was limited by the small number of cases. Surgeons may have encouraged earlier discharge of the patients in the LDG group. A high-volume, prospective, and randomized study is needed to confirm our findings.
According to the Japanese gastric cancer treatment guidelines, laparoscopic surgery is one of the most reliable treatments for early gastric cancer. It is important to determine whether elderly patients with advanced gastric cancer can benefit from laparoscopic surgery. Our results demonstrated that LDG for advanced gastric cancer in patients older than 75 years was associated with lesser intraoperative blood loss and shorter hospital stays than ODG, with no differences in survival. Patient age alone should not rule out the feasibility of either LDG or ODG.
Declarations
AcknowledgmentsWe are grateful to the study participants and their caregivers. The authors thank participating surgeons from the Bell Land General Hospital. We would like to thank Editage (www.editage.jp) for English language editing.
Authors’ contributionsDesigned the study and acquisition, analysis, and interpretation of the data: Yuu K
Collected the data: Yuu K, Tsuchihashi K, Toyoda S, Kawasaki M
Supervised this study: Kameyama M
Availability of data and materialsNot applicable.
Financial support and sponsorshipNone.
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateThis study was approved by the institutional review board of the Bell Land General Hospital. The study protocol conformed to the principles set in the Declaration of Helsinki and its later amendments. Written informed consent was obtained from all patients before the operation.
Consent for publicationWritten informed consent was obtained from all patients before the operation.
Copyright© The Author(s) 2019.
REFERENCES
1. Frasson M, Braga M, Vignali A, Zuliani W, Di Carlo V. Benefits of laparoscopic colorectal resection are more pronounced in elderly patients. Dis Colon Rectum 2008;51:296-300.
2. Endo S, Dousei T, Yoshikawa Y, Hatanaka N, Kamiike W, et al. Prognosis of gastric carcinoma patients aged 85 years or older who underwent surgery or who received best supportive care only. Int J Clin Oncol 2013;18:1014-9.
3. Kim EJ, Seo KW, Yoon KY. Laparoscopy-assisted distal gastrectomy for early gastric cancer in the elderly. J Gastric Cancer 2012;12:232-6.
4. Polanczyk CA, Marcantonio E, Goldman L, Rohde LE, Orav J, et al. Impact of age on perioperative complications and length of stay in patients undergoing noncardiac surgery. Ann Intern Med 2001;134:637-43.
5. Monson K, Litvak DA, Bold RJ. Surgery in the aged population: surgical oncology. Arch Surg 2003;138:1061-7.
6. Story DA. Postoperative complications in elderly patients and their significance for long-term prognosis. Curr Opin Anaesthesiol 2008;21:375-9.
7. Kim MC, Jung GJ, Kim HH. Morbidity and mortality of laparoscopy-assisted gastrectomy with extraperigastric lymph node dissection for gastric cancer. Dig Dis Sci 2007;52:543-8.
8. Kitano S, Shiraishi N, Fujii K, Yasuda K, Inomata M, et al. A randomized controlled trial comparing open vs laparoscopy-assisted distal gastrectomy for the treatment of early gastric cancer: an interim report. Surgery 2002;131:S306-11.
9. Mochiki E, Nakabayashi T, Kamimura H, Haga N, Asao T, et al. Gastrointestinal recovery and outcome after laparoscopy-assisted versus conventional open distal gastrectomy for early gastric cancer. World J Surg 2002;26:1145-9.
10. Kim MG, Kim HS, Kim BS, Kwon SJ. The impact of old age on surgical outcomes of totally laparoscopic gastrectomy for gastric cancer. Surg Endosc 2013;27:3990-7.
11. Park HA, Park SH, Cho SI, Jang YJ, Kim JH, et al. Impact of age and comorbidity on the short-term surgical outcome after laparoscopy-assisted distal gastrectomy for adenocarcinoma. Am Surg 2013;79:40-8.
12. Japanese Gastric Cancer Association. Japanese classification of gastric carcinoma: 3rd English edition. Gastric Cancer 2011;14:101-12.
13. Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chron Dis 1987;40:373-83.
14. Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 2004;240:205-13.
15. Kanda Y. Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transplant 2013;48:452-8.
16. Kawamura H, Yokota R, Homma S, Kondo Y. Comparison of respiratory function recovery in the early phase after laparoscopy-assisted gastrectomy and open gastrectomy. Surg Endosc 2010;24:2739-42.
17. Inokuchi M, Kojima K, Kato K, Motoyama K, Sugita H, et al. Feasibility of laparoscopy-assisted gastrectomy for patients with chronic obstructive pulmonary disease. Surg Endosc 2013;27:2102-9.
18. Fletcher CM. The clinical diagnosis of pulmonary emphysema; an experimental study. Proc R Soc Med 1952;45:577-84.
19. Evers BM, Townsend CM Jr, Thompson JC. Organ physiology of aging. Surg Clin North Am 1994;74:23-39.
20. Japanese gastric cancer treatment guidelines (ver.3). Tokyo: Kanehara; 2010.
21. Mayol J, Martinez-Sarmiento J, Tamayo FJ, Fernández-Represa JA. Complications of laparoscopic cholecystectomy in the ageing patient. Age Ageing 1997;26:77-81.
22. Turrentine FE, Wang H, Simpson VB, Jones RS. Surgical risk factors, morbidity, and mortality in elderly patients. J Am Coll Surg 2006;203:865-77.
23. Odeberg S, Ljungqvist O, Svenberg T, Gannedahl P, Bäckdahl M, et al. Haemodynamic effects of pneumoperitoneum and the influence of posture during anaesthesia for laparoscopic surgery. Acta Anaesthesiol Scand 1994;38:276-83.
24. Rauh R, Hemmerling TM, Rist M, Jacobi KE. Influence of pneumoperitoneum and patient positioning on respiratory system compliance. J Clin Anesth 2001;13:361-5.
25. Gutt CN, Oniu T, Mehrabi A, Schemmer P, Kashfi A, et al. Circulatory and respiratory complications of carbon dioxide insufflation. Dig Surg 2004;21:95-105.
26. Lu J, Huang CM, Zheng CH, Li P, Xie JW, et al. Short- and long-term outcomes after laparoscopic versus open total gastrectomy for elderly gastric cancer patients: a propensity score-matched analysis. J Gastrointest Surg 2015;19:1949-57.
27. Usui S, Yoshida T, Ito K, Hiranuma S, Kudo SE, et al. Laparoscopy-assisted total gastrectomy for early gastric cancer: comparison with conventional open total gastrectomy. Surg Laparosc Endosc Percutan Tech 2005;15:309-14.
28. Schweickert WD, Pohlman MC, Pohlman AS, Nigos C, Pawlik AJ, et al. Early physical and occupational therapy in mechanically ventilated, critically ill patients: a randomised controlled trial. Lancet 2009;373:1874-82.
29. Avidan MS, Fritz BA, Maybrier HR, Muench MR, Escallier KE, et al. The prevention of delirium and complications associated with surgical treatments (PODCAST) study: protocol for an international multicentre randomised controlled trial. BMJ Open 2014;4:e005651.
30. Hu Y, Ying M, Huang C, Wei H, Jiang Z, et al. Oncologic outcomes of laparoscopy-assisted gastrectomy for advanced gastric cancer: a large-scale multicenter retrospective cohort study from China. Surg Endosc 2014;28:2048-56.
31. Wu CW, Lo SS, Shen KH, Hsieh MC, Lui WY, et al. Surgical mortality, survival, and quality of life after resection for gastric cancer in the elderly. World J Surg 2000;24:465-72.
32. Kitamura K, Sugimachi K, Saku M. Evaluation of surgical treatment for patients with gastric cancer who are over 80 years of age. Hepatogastroenterology 1999;46:2074-80.
33. Zheng L, Lu L, Jiang X, Jian W, Liu Z, et al. Laparoscopy-assisted versus open distal gastrectomy for gastric cancer in elderly patients: a retrospective comparative study. Surg Endosc 2016;30:4069-77.
34. Park SH, Mok YJ, Kim JH, Park SS, Kim SJ, et al. Clinical significance of gastric outlet obstruction on the oncologic and surgical outcomes of radical surgery for carcinoma of the distal stomach. J Surg Oncol 2009;100:215-21.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Yuu K, Tsuchihashi K, Toyoda S, Kawasaki M, Kameyama M. Laparoscopic vs. open distal gastrectomy for advanced gastric cancer in elderly patients: a retrospective study. Mini-invasive Surg 2019;3:6. http://dx.doi.org/10.20517/2574-1225.2018.73
AMA Style
Yuu K, Tsuchihashi K, Toyoda S, Kawasaki M, Kameyama M. Laparoscopic vs. open distal gastrectomy for advanced gastric cancer in elderly patients: a retrospective study. Mini-invasive Surgery. 2019; 3: 6. http://dx.doi.org/10.20517/2574-1225.2018.73
Chicago/Turabian Style
Yuu, Ken, Kurumi Tsuchihashi, Sho Toyoda, Masayasu Kawasaki, Masao Kameyama. 2019. "Laparoscopic vs. open distal gastrectomy for advanced gastric cancer in elderly patients: a retrospective study" Mini-invasive Surgery. 3: 6. http://dx.doi.org/10.20517/2574-1225.2018.73
ACS Style
Yuu, K.; Tsuchihashi K.; Toyoda S.; Kawasaki M.; Kameyama M. Laparoscopic vs. open distal gastrectomy for advanced gastric cancer in elderly patients: a retrospective study. Mini-invasive. Surg. 2019, 3, 6. http://dx.doi.org/10.20517/2574-1225.2018.73
About This Article
Special Issue
Copyright
Data & Comments
Data

 Cite This Article 4 clicks
Cite This Article 4 clicks










Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.