Totally laparoscopic total gastrectomy: challenging but feasible: a single center case series
Abstract
Aim: To report the initial monocentric experience of totally laparoscopic total gastrectomy, assessing its feasibility and safety, especially relating to the challenging step of esophago-jejunal (E-J) reconstruction.
Methods: All consecutive patients, underwent laparoscopic total gastrectomy for gastric cancer with curative intent, between January 2017 and June 2018 at our institution, were considered. Data of the selected patients was retrieved from a prospectively collected database. Short and long term outcomes were analyzed.
Results: Ten patients underwent totally laparoscopic total gastrectomy with D2 lymphadenectomy and 4 of these had received preoperative chemotherapy; Two patients also received the lymphadenectomy of the station 10. E-J reconstruction consisted of hemi-double stapling technique with transorally inserted anvil in 1 case, side-to-side overlap anastomosis in 5 cases and end-to-side anastomosis in 4 cases. One patient experienced intraoperative complications needing conversion to laparotomy. Seven patients experienced postoperative complications, three of these were severe according to Dindo-Clavien classification. All the specimens had free proximal resection margins with R0 resection in all the cases. Average postoperative length of hospital stay was 10 days and no patients died during hospitalization. Median overall survival and disease-free survival were 15.5 and 12.5 months respectively.
Conclusion: Totally laparoscopic total gastrectomy is a feasible and safe option in the treatment of gastric cancer. The choice about the type of E-J reconstruction should be based on the single patient’s features and on the dexterity of the surgeon who should be able to perform more than one option for a tailored approach.
Keywords
Introduction
Gastric cancer is the second leading cause of cancer-related mortality worldwide[1]. Although relevant improvements in medical oncology, radical surgery still remains the mainstay of curative treatment[2]. Laparoscopic distal gastrectomy reached a wide diffusion in the last decades, before the treatment of early gastric cancer, and then treating advanced gastric cancer too[3], especially in Eastern countries, demonstrating clear advantages in terms of intraoperative bleeding, length of hospital stay, restoration of bowel function and incidences of minor postoperative complications in comparison with open surgery[4]. Looking at these results, laparoscopic technique has been gradually adopted, including total gastrectomy, confirming the short-term benefits as compared to open technique[5]. On the other hand, laparoscopic total gastrectomy is considered a very demanding procedure, due to the complexity of many steps like omentectomy and lymphadenectomy; however the main reason of complexity is the technical difficulty of esophago-jejunal reconstruction[6].
In order to overcome this complexity, many techniques have been proposed, ranging from hybrid anastomosis by mini-laparotomy, to different kinds of totally laparoscopic ones using a circular or linear stapler, without a clear superiority of one above the others[7]. Furthermore the majority of data on this topic comes from Eastern countries and from case series, making the debate still open[8]. In this scenario, we reported our experience of totally laparoscopic total gastrectomy.
Methods
All consecutive patients who underwent laparoscopic total gastrectomy for gastric cancer with curative intent, between January 2017 and June 2018 at our institution, were considered. Patients with esophago-gastric junction cancer, with evidence of metastatic disease and/or underwent surgery with palliative intent were excluded.
Exclusion criteria for laparoscopic treatment of gastric cancer were: tumor involvement of adjacent organs, T4a tumor at endoscopic ultrasonography (EUS), tumor located in the greater curvature, preoperative evidence of bulky lymph-nodes, anesthesiologic contraindications to pneumoperitoneum, history of other surgery for gastric cancer, previous laparotomies for major upper abdominal surgery.
All the patients underwent preoperative complete workup consisting of esophago-gastro-duodenoscopy with biopsy, gastric EUS, total body contrast-enhanced computed tomography (ce-CT). Tumor staging was performed according to American Joint Committee on Cancer (AJCC) tumor/node/metastasis classification and staging systems for gastric cancer 8th edition[9].
In all the patients with more than T2 N0 tumors, a perioperative chemotherapy was performed, according to age, comorbidity and performance status and it was followed by a re-staging of the disease. Complications were defined according to the Dindo-Clavien classification; severe complications were considered those of grade > 2[10]. Data of the selected patients was retrieved from a prospectively collected database; all the gathered data was recorded on an electronic spreadsheet and analysed using commercially available software, SPSS 18.0 (IBM, Armonk, NY).
Perioperative management and technical details
All patients received low molecular weight heparin starting the evening before the surgery, 2 g cefazolin 15 min before the skin incision, and 7-10 days preoperatively immunonutrition with 2 brick/day of impact oral.
Patients were placed in a supine position, with legs apart, with the first surgeon positioned between the legs and one assistant on each side of the patient’s abdomen. An open technique according to Hasson was used for the first trocar insertion in suvra-humbilical position; other trocars were placed, 2 on each side of the umbilicus, drawing a “smile” shape line; of these, three were 5 mm and one (on the left hemi-clavear line) 12 mm. A 30 degree scope was always used, with a carbon dioxide 12-14 mmHg pneumoperitoneum.
An exploration of the peritoneal cavity was also performed in order to exclude carcinomatosis and a sample of peritoneal liquid was always obtained for cytological examination.
Coloepiploic detachment and complete omentectomy was the first step of the operation with section of left gastroepiploic vessels at their origin, removing lymph-nodes of stations 4sb and 4sa, section of short gastric vessels, followed by right gastroepiploic vessels section, removing stations 5, 6 and 4d.
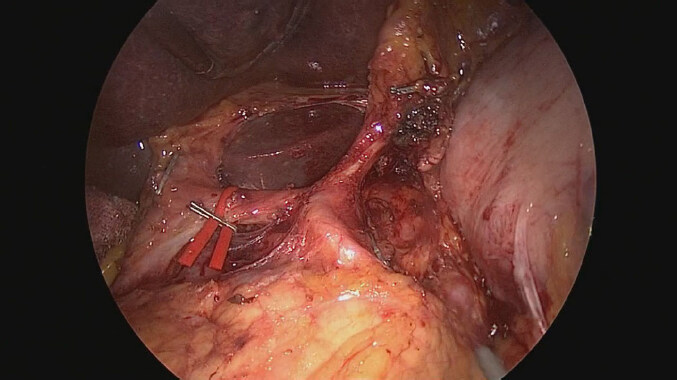
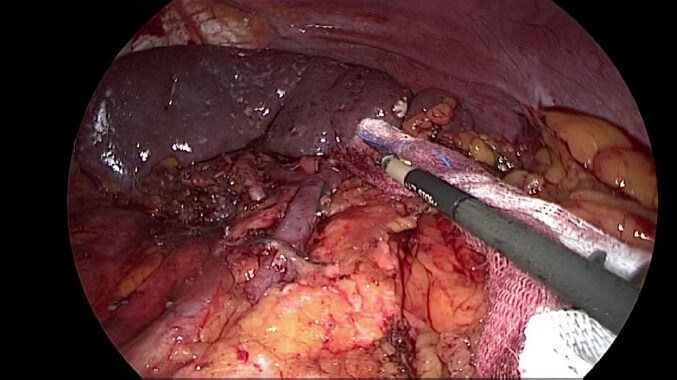
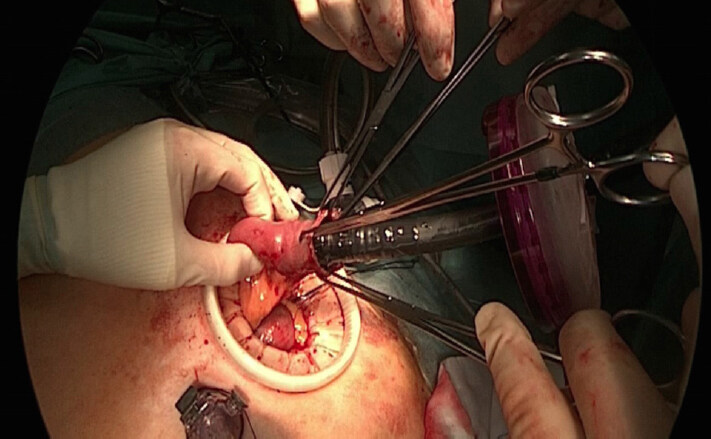
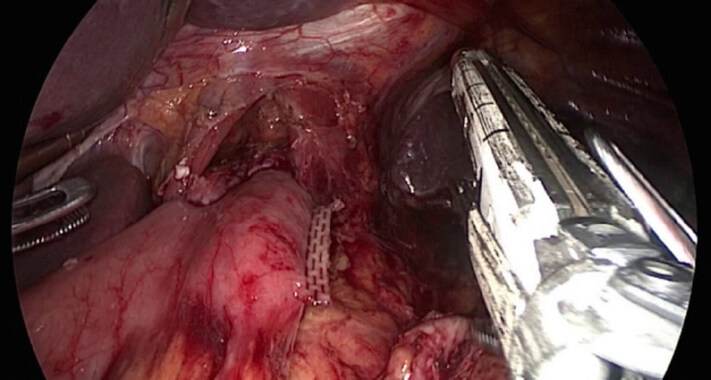
Division of the duodenum was performed 2-3 cm distal to pylorus, using 60 mm tristaple Endo Gia reinforced with polyglicolic acid (bioabsorbable staple line reinforcement, GORE SEAMGUARDTM). Hepatic pedicle was cleared removing lymph-nodes of the station 12a untill the portal vein became visible and right gastric artery was sectioned. Stomach was pushed upward with better exposition of the suprapancreatic region; lymphadenectomy of stations 7-8a-9-11p-11d was performed and left gastric vessels were divided using clips [Figure 1]. Laimer-Bertelli membrane was divided and cardial branches and of the vagus nerves sectioned, removing lymph-nodes of the stations 1, 2 and 3. Dissection of the station 10 was performed only in the presence of enlarged lymph-nodes at splenic hylum [Figure 2].
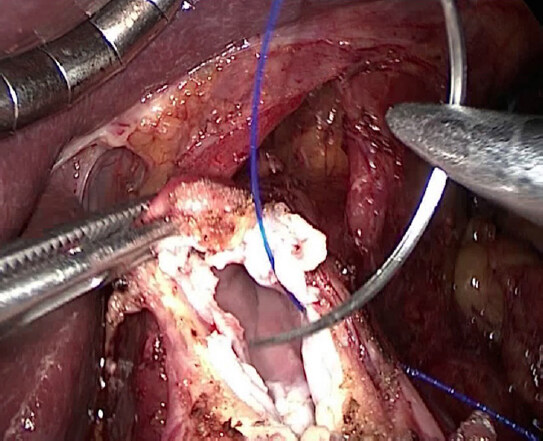
Roux-en-Y reconstruction was always done using the transmesocolic route and jejuno-jejunal anastomosis was perforemed with the same technique (isoperistaltic side-to-side mechanical anastomosis using 45 mm linear stapler) in all the patients. According to patients’ characteristics and surgeon preference, 3 techniques were used for esophago-jejunal (E-J) anastomosis: hemi-double-stapling (HDS) technique using the transorally inserted anvil (OrVilTM)[11], modified side-to-side (S-S) overlap anastomosis according to Inaba[12], and modified end-to-side (E-S) anastomosis. For the last one, jejunal loop was always marked with a pen about 20 cm distally to the Treitz ligament and sectioned using a 45 mm linear stapler after it was passed through the mesocolic breach. The anterior hemi-circumference of the distal esophagus was sectioned with monopolar coagulation or an ultrasound device and a hemi hand-sewn purse-string, using polypropylene 2/0 suture, was placed [Figure 3].
Figure 3. Placement of the hemi hand-sewn purse-string, using polypropylene 2/0 suture, on the anterior esophageal circumference
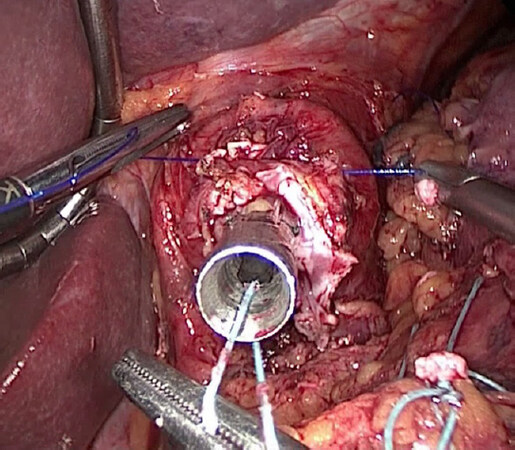
After a stitch was placed on its edge, the anvil was introduced in the peritoneal cavity through the suvra-humbilical port, and inserted in the esophagus under laparoscopic vision. The remaining esophageal circumference was sectioned and the hand-sewn purse-string completed, using the stitch on the anvil edge to pull it in the correct position [Figure 4]. The specimen was extracted through mini-laparotomy on the left hemi-clavear trocar; the same mini-laparotomy was used to place the circular stapler in the previously sectioned jejunal loop and to reintroduce it in the peritoneal cavity restoring the pneumoperitoneum using a specialized wound-sealing device (GelPOINT Access Platforms, Applied MedicalTM) [Figure 5]. The E-S E-J anastomosis was performed using 25 mm circular stapler and the jejunal loop extremity was then sectioned using linear 45 mm stapler [Figure 6]. In all the patients an external close suction drain (type Jackson Pratt) was positioned via the right subcostal 5 mm port and positioned posteriorly to the E-J anastomosis. All the patients resumed oral idric intake on 1st postoperative day and semi liquid diet on 2nd postoperative day, when tolerated. A ce-CT of thorax and abdomen with oral hydrosoluble contrast examination was always performed on 6th postoperative day. After hospital discharge, follow-up was continued in outpatient settings every six months.
Figure 5. After specimen extraction, the mini-laparotomy on the left hemi-clavear trocar was used to place the circular stapler in the previously sectioned jejunal loop and to reintroduce it in the peritoneal cavity restoring the pneumoperitoneum using a specialized wound-sealing device
Results
Between January 2017 and June 2018 at our institution, 28 patients underwent surgery for middle-upper third gastric cancer with curative intent; among these: 4 had previous gastric surgery for cancer and received degastrogastrectomy, 4 underwent associated transhiatal distal esophagectomy, 1 underwent upper polar gastrectomy. The remaining 19 underwent total gastrectomy; 9 of these received laparotomic treatment because of anesthesiological contraindications to laparoscopy (2 patients), tumor involvement of adjacent organs (4 patients), evidence of bulky nodes (1 patient), evidence of T4a tumor (1 patient), or tumor located on the greater curvature (1 patient). 10 patients underwent totally laparoscopic total gastrectomy with D2 lymphadenectomy and 4 of these had received preoperative chemotherapy; in 2 cases the lymphadenectomy of the station 10 was performed. Demographic and preoperative features of these patients are reported in Table 1. After an uneventful resective phase in all patients, 1 of these received E-J anastomosis using HDS technique with transorally inserted anvil, 5 received S-S overlap anastomosis and 4 underwent E-S anastomosis. Only one patient experienced intraoperative complications; the only case reported was related to an esophageal injury during S-S E-J anastomosis needing the only conversion to laparotomy of the series; after conversion an E-S E-J anastomosis with a hand-sewn purse-string was done without further complications; other intraoperative details are reported in Table 2. Although no 30 day fatal events occurred, 7 patients experienced postoperative complications, 3 of these were severe according to Dindo-Clavien classification[10]; they consisted of ileus due to internal hernia, and abdominal abscess in splenic fossa needing reoperation; the last one was the only E-J anastomotic leakage and occurred in a patient who underwent laparoscopic E-S anastomosis and treated with endoscopic transanastomotic stent positioning. At pathological examination all the specimens had free proximal resection margins and all the patients received R0 resection; average numbers of harvested lymph-nodes was 29. Clinical staging was confirmed in 5 patients; on the other hand, 2 patients were under-staged and 3 over-staged; this miss-staging didn’t affect the curative intent of surgery and no peritoneal carcinosis was detected at laparoscopy.
Patients’ characteristics
| Number of patients | 10 |
| Sex ratio (M/F) | 6/4 |
| Age (year)* | 76 (60-93) |
| BMI (kg/m2)* | 24 (19-32) |
| ASA score 1/2/3 | 1/6/3 |
| clinical T1/T2/T3# | 1/2/7 |
| clinical EGC/AGC ratio | 1/9 |
| clinical N0/N+# | 3/7 |
| clinical stage I/IIa/IIb/III# | 1/2/2/5 |
| Preoperative CT | 4 |
Intraoperative outcomes
| Duration of surgery (min)* | 369 (275-440) |
|---|---|
| Type of anastomosis: | |
| HDS technique with transorally inserted anvil | 1 |
| S-S overlap anastomosis | 5 |
| E-S anastomosis | 4 |
| Intraoperative complication | 1 |
| Associated procedures | 6 |
| Conversion to open surgery | 1 |
Average postoperative length of hospital stay was 10 days and no patients died during hospitalization. With a minimum follow-up of 6 months, median overall survival (OS) and disease-free survival (DFS) were 15.5 and 12.5 months respectively. All the patients included in perioperative chemotherapy program were able to complete the post operative treatment. Postoperative outcomes are described in Table 3.
Postoperative outcomes
| Length of hospital stay (day)* | 10 (8-58) |
| Overall complications | 7 |
| Severe complications | 3 |
| Reoperations | 2 |
| Anastomotic leakage | 1 |
| Anastomotic stenosis | 0 |
| Abdominal abscess | 1 |
| POPF | 0 |
| Duodenal stump leak | 0 |
| Wound infection | 0 |
| Ileus caused by internal hernia | 1 |
| Number of LNs harvested§ | 29 (15-38) |
| Number of LNs positive§ | 5 (0-22) |
| Tumor dimension (mm) | 49 (17-130) |
| Pathological T1b/T2/T3/T4a# | 1/1/6/2 |
| Pathological N0/N1/N2/N3a/N3b | 3/1/1/4/1 |
| Pathological staging Ib/IIa/IIIa/IIIb/IIIc# | 2/2/1/4/1 |
| Median OS* (months) | 15.5 (6-32) |
| Median DFS* (months) | 12.5 (6-28) |
Discussion
Laparoscopic surgery is a valid option for the treatment of gastric cancer; although only the minor part of the data about it comes from randomized clinical trials (RCTs)[13,14]. A large number of results from non RCTs and case series assessed its safety and feasibility[4,15], however the majority of the reported data refers to distal gastrectomy[16] and only a few parts of the ongoing RCTs include total gastrectomy[17-19].
Nevertheless it gained wide diffusion in Eastern countries, with a slow adoption in Europe and USA too[11,20], showing less blood loss, fewer analgesic uses, earlier passage of flatus, quicker resumption of oral intake, earlier hospital discharge, and reduced postoperative morbidity, with longer operative time, in comparison with open total gastrectomy[5]. Our data, obtained from a non selected series of European older adults, was consistent with these evidences, reporting 3 cases of severe adverse events, 2 of these requiring reoperation; 1 of these was due to an internal hernia and since that occurance we always have sutured the jejunal mesentery without occurance of further cases.
Some critical aspects of this procedure however still make it open to debate, one of these being the possibility to perform a correct lymphadenectomy, especially to dissect the station 10. A large meta-analysis, comparing open and laparoscopic total gastrectomy, reported no statistical differences between the two techniques in terms of lymph-nodes clearance, 5-year OS and DFS, and free proximal resection margins, confirming their same oncological safety and adequacy[5]. This data was confirmed by another meta-analysis, including totally laparoscopic total gastrectomy only, reporting no difference in the number of the harvested lymph-nodes[21]. Furthermore a recent RCT[22] reported an incidence of positive lymph-nodes in the station 10 of 2.4%, in a cohort of well selected patients, all candidates to total gastrectomy, concluding that, for these kind of patients, the station 10 lymphadenectomy is not mandatory; however in case of macroscopic lymph-nodes at splenic hilum it seems possible to perform a laparoscopic spleen-preserving dissection[23]. Our small series confirmed this data with a mean number of lymph-nodes harvested of 29; D2 lymphadenectomy and complete omentectomy were always performed in all the patients and they didn’t cause intraoperative nor postoperative complications; 2 patients needed, in addition, the spleen-preserving lymphadenectomy of the station 10 for slightly enlarged, suspicious, lymph-nodes without further morbidity.
Therefore the major concern of laparoscopic total gastrectomy seems related to the E-J reconstruction. The first attempt to overcome this obstacle was to perform a midline mini-laparotomy for a hybrid approach; however several authors reported important limits of this approach due to the difficulty to use a purse-string instrument in a very narrow space, and to place a suture without clear visualization[24,25]. Actually various totally laparoscopic techniques are used, based on circular or linear staplers[7]; some authors achieved excellent results independently from the technique, without a clear superiority of a particular model[1], even if leakage and stenosis seemed to occur more frequently using circular stapler compared to linear ones[8]. However the incidence of anastomotic-related complications widely varied among the studies with anastomotic leakage and stricture rate of 3.5% (0.9%-8.5%) and 2.2% (0%-9%) respectively[1]. Differences between laparoscopic total gastrectomies with open ones were not found, neither regarding anastomotic-related complications, with a slightly, but not significantly, disadvantage of laparoscopy in term of stenosis[26]. The feasibility and good outcomes of totally laparoscopic reconstruction were supported by our data; in the majority of patients, anastomosis was performed laparoscopically, under pneumoperitoneum, with a good visualization; just in one case an esophageal disruption occurred during linear stapler insertion needing laparotomic conversion to re-do the anastomosis. With a mean follow up of 16 month, only 1 anastomotic-related complication occurred: anastomotic leakage diagnosed at imaging on 6th postoperative day and treated by means of endoscopically positioned stent with good recovery. As reported in literature, overlap technique is quite easy to perform and not demanding a long time; in addition it seemed to have lower incidence of anastomotic stenosis compared to Orvill technique[27]; on the other hand it doesn’t allow ability to check for malignancy with frozen section the esophageal margin[6] and can be very challenging in obese patients with thickened and fixed mesentery, as the patient in our series who experienced esophageal injury. For patients with these anthropometric features, we used a modified E-S double stapling technique using 25 mm circular stapler, introduced in the peritoneal cavity via a wound sealing device in the place of the left hemi-clavear trocar, reproducing the same familial anastomosis usually used in the open total gastrectomy. This technique, although perceived as more time-consuming and technically challenging, allowed to perform a tension-free anastomosis in obese patients too, overcoming the problem of the deep and dorsal position of the anastomotic site and the hindering visualization and manipulation.
Small sample size, retrospective and non comparative analysis constitute the main limits of this study, and firm conclusions cannot be drawn, but it adds to a still lacking amount of literature, especially in Western countries, some results confirming the feasibility and the safety of totally laparoscopic total gastrectomy in the hands of experienced surgeons with a steep learning curve.
In conclusion, totally laparoscopic total gastrectomy is a feasible option in the treatment of gastric cancer. The choice about the method for E-J reconstruction should be based on the individual patient’s features and on the dexterity of the surgeon, which should be able to perform more than one option for a tailored approach.
Declarations
Authors’ contributionsMade substantial contributions to conception and design of the study: Mazzola M, Gualtierotti M, De Martini P, Ferrari G
Performed data analysis and interpretation: Mazzola M, Gualtierotti M
Performed data acquisition: Morini L, Achilli P, Zironda A
Availability of data and materialsNot appliable.
Financial support and sponsorshipNone.
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Author(s) 2019.
REFERENCES
1. Okabe H, Tsunoda S, Tanaka E, Hisamori S, Kawada H, et al. Is laparoscopic total gastrectomy a safe operation? A review of various anastomotic techniques and their outcomes. Surg Today 2015;45:549-58.
2. Coburn N, Cosby R, Klein L, Knight G, Malthaner R, et al. Staging and surgical approaches in gastric cancer: A clinical practice guideline. Curr Oncol 2017;24:324-31.
3. Inaki N, Etoh T, Ohyama T, Uchiyama K, Katada N, et al. A Multi-institutional, Prospective, Phase II Feasibility Study of Laparoscopy-Assisted Distal Gastrectomy with D2 Lymph Node Dissection for Locally Advanced Gastric Cancer (JLSSG0901). World J Surg 2015;39:2734-41.
4. Best LM, Mughal M, Gurusamy KS. Laparoscopic versus open gastrectomy for gastric cancer. Cochrane Database Syst Rev 2016;3:CD011389.
5. Wang W, Zhang X, Shen C, Zhi X, Wang B, et al. Laparoscopic versus open total gastrectomy for gastric cancer: An updated meta-analysis. PLoS One 2014;9:e88753.
6. Treitl D, Hochwald SN, Bao PQ, Unger JM, Ben-David K. Laparoscopic Total Gastrectomy with D2 Lymphadenectomy and Side-to-Side Stapled Esophagojejunostomy. J Gastrointest Surg 2016;20:1523-9.
7. Wei G, Zheng J, Li Y. Reconstruction after LATG : technical tips and pitfalls. Transl Gastroenterol Hepatol 2017;2:41.
8. Umemura A, Koeda K, Sasaki A, Fujiwara H, Kimura Y, et al. Totally laparoscopic total gastrectomy for gastric cancer: Literature review and comparison of the procedure of esophagojejunostomy. Asian J Surg 2015;38:102-12.
9. Amin MB. AJCC Cancer Staging System, 8th Edition : UPDATE. Am Jt Comm Cancer 2013. Avaliable from: http://cancerstaging.org/About/Pages/8th-Edition.aspx. [Last accessed on 9 Apr 2019].
10. Dindo D, Demartines N, Clavien PA. Classification of Surgical Complications. Ann Surg 2004;240:205-13.
11. Lafemina J, Viñuela EF, Schattner MA, Gerdes H, Strong VE. Esophagojejunal reconstruction after total gastrectomy for gastric cancer using a transorally inserted anvil delivery system. Ann Surg Oncol 2013;20:2975-83.
12. Inaba K, Satoh S, Ishida Y, Taniguchi K, Isogaki J, et al. Overlap method: Novel intracorporeal esophagojejunostomy after laparoscopic total gastrectomy. J Am Coll Surg 2010;211:225-9.
13. Kitano S, Shiraishi N, Fujii K, Yasuda K, Inomata M, et al. A randomized controlled trial comparing open vs laparoscopy-assisted distal gastrectomy for the treatment of early gastric cancer: An interim report. Surgery 2002;131:306-11.
14. Lee JH, Han HS, Lee JH. A prospective randomized study comparing open vs laparoscopy-assisted distal gastrectomy in early gastric cancer: early results. Surg Endosc 2005;19:168-73.
15. Lin JX, Huang CM, Zheng CH, Li P, Xie JW, et al. Surgical outcomes of 2041 consecutive laparoscopic gastrectomy procedures for gastric cancer: A large-scale case control study. PLoS One 2015;10:e0114948.
16. Kim YW, Yoon HM, Yun YH, Nam BH, Eom BW, et al. Long-term outcomes of laparoscopy-assisted distal gastrectomy for early gastric cancer: Result of a randomized controlled trial (COACT 0301). Surg Endosc Other Interv Tech 2013;27:4267-76.
17. Straatman J, van der Wielen N, Cuesta MA, Gisbertz SS, Hartemink KJ, et al. Surgical techniques, open versus minimally invasive gastrectomy after chemotherapy (STOMACH trial): study protocol for a randomized controlled trial. Trials 2015;16:123.
18. Haverkamp L, Brenkman HJ, Seesing MF, Gisbertz SS, van Berge Henegouwen MI, et al. Laparoscopic versus open gastrectomy for gastric cancer, a multicenter prospectively randomized controlled trial (LOGICA-trial). BMC Cancer 2015;15:556.
19. Hyung WJ, Yang HK, Han SU, Lee YJ, Park JM, et al. A feasibility study of laparoscopic total gastrectomy for clinical stage I gastric cancer: a prospective multi-center phase II clinical trial, KLASS 03. Gastric Cancer 2019;22:214-22.
20. Kostakis ID, Alexandrou A, Armeni E, Damaskos C, Kouraklis G, et al. Comparison Between Minimally Invasive and Open Gastrectomy for Gastric Cancer in Europe: A Systematic Review and Meta-analysis. Scand J Surg 2017;106:3-20.
21. Malczak P, Torbicz G, Rubinkiewicz M, Gajewska N, Sajuk N, et al. Comparison of totally laparoscopic and open approach in total gastrectomy with D2 lymphadenectomy - systematic review and meta-analysis. Cancer Manag Res 2018;10:6705-14.
22. Sano T, Sasako M, Mizusawa J, Yamamoto S, Katai H, et al. Randomized Controlled Trial to Evaluate Splenectomy in Total Gastrectomy for Proximal Gastric Carcinoma. Ann Surg 2017;265:277-83.
23. Tu RH, Li P, Xie JW, Wang JB, Lin JX, et al. Development of lymph node dissection in laparoscopic gastrectomy: safety and technical tips. Transl Gastroenterol Hepatol 2017;2:23.
24. Bo T, Peiwu Y, Feng Q, Yongliang Z, Yan S, et al. Laparoscopy-Assisted vs. Open Total Gastrectomy for Advanced Gastric Cancer: Long-Term Outcomes and Technical Aspects of a Case-Control Study. J Gastrointest Surg 2013;17:1202-8.
25. Jeong O, Ryu SY, Zhao XF, Jung MR, Kim KY, et al. Short-term surgical outcomes and operative risks of laparoscopic total gastrectomy (LTG) for gastric carcinoma: Experience at a large-volume center. Surg Endosc 2012;26:3418-25.
26. Inokuchi M, Otsuki S, Fujimori Y, Sato Y, Nakagawa M, et al. Systematic review of anastomotic complications of esophagojejunostomy after laparoscopic total gastrectomy. World J Gastroenterol 2015;21:9656-65.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Mazzola M, Gualtierotti M, De Martini P, Bertoglio CL, Morini L, Achilli P, Zironda A, Ferrari G. Totally laparoscopic total gastrectomy: challenging but feasible: a single center case series. Mini-invasive Surg 2019;3:12. http://dx.doi.org/10.20517/2574-1225.2019.05
AMA Style
Mazzola M, Gualtierotti M, De Martini P, Bertoglio CL, Morini L, Achilli P, Zironda A, Ferrari G. Totally laparoscopic total gastrectomy: challenging but feasible: a single center case series. Mini-invasive Surgery. 2019; 3: 12. http://dx.doi.org/10.20517/2574-1225.2019.05
Chicago/Turabian Style
Mazzola, Michele, Monica Gualtierotti, Paolo De Martini, Camillo Leonardo Bertoglio, Lorenzo Morini, Pietro Achilli, Andrea Zironda, Giovanni Ferrari. 2019. "Totally laparoscopic total gastrectomy: challenging but feasible: a single center case series" Mini-invasive Surgery. 3: 12. http://dx.doi.org/10.20517/2574-1225.2019.05
ACS Style
Mazzola, M.; Gualtierotti M.; De Martini P.; Bertoglio CL.; Morini L.; Achilli P.; Zironda A.; Ferrari G. Totally laparoscopic total gastrectomy: challenging but feasible: a single center case series. Mini-invasive. Surg. 2019, 3, 12. http://dx.doi.org/10.20517/2574-1225.2019.05
About This Article
Special Issue
Copyright
Data & Comments
Data

 Cite This Article 3 clicks
Cite This Article 3 clicks










Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.