Minimally invasive surgery for gallbladder cancer at an expert center
Abstract
In this article, we reviewed the techniques and outcomes of minimally invasive surgery for gallbladder cancer performed at an expert center. The techniques of laparoscopic extended cholecystectomy with the short- and long-term outcomes at our center were described. The short- and long-term survival outcomes of laparoscopic extended cholecystectomy are comparable to open surgery. Laparoscopic surgery is a safe, effective alternative for open surgery in the treatment of gallbladder cancer. The benefits of robotic surgery should be proven with further research.
Keywords
INTRODUCTION
The treatment of gallbladder cancer (GBC) can be variable depending on the stage of disease. A variety of operations are performed, from simple cholecystectomy performed for T1a cancers to extended right hemihepatectomy and bile duct resection performed for more advanced cancers. There are several decisions for the surgeon to make[1]. The first decision to be made is whether to proceed with surgery. The role of staging laparoscopy to aid this decision has been reported[2]. Thereafter, the operator should decide whether to perform lymphadenectomy and its extent, whether to perform a liver wedge resection or a formal hepatectomy, and whether to perform a bile duct resection or not.
In the era of minimally invasive surgery (MIS), the treatment options for the surgeon are even more complicated. Currently, open surgery, laparoscopic surgery, and robotic surgery are all being performed[3]. Therefore, it is necessary to evaluate the merits and demerits of these operation methods.
We have started a prospective study on “the laparoscopic approach for early GBC” in 2004, and have experienced a significant number of cases since. In this article, we review our history of MIS for GBC.
STEP BY STEP ADOPTION OF LAPAROSCOPIC SURGERY FOR GBC
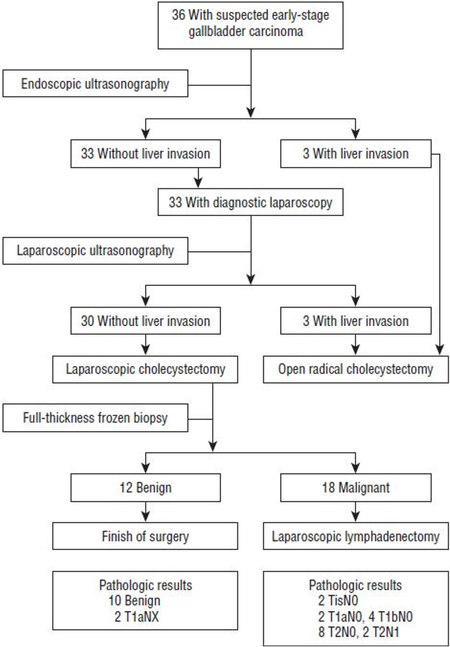
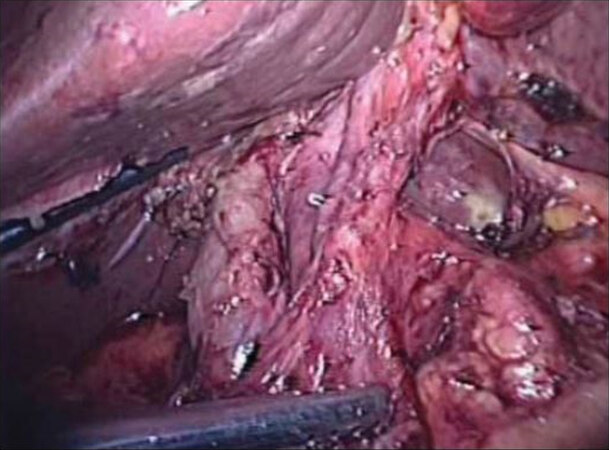
Laparoscopic surgery for GBC was contraindicated for a long time, although cholecystectomy was the first laparoscopic surgery in the field of general surgery. With experience of MIS in various fields, we came to believe that laparoscopic surgery is beneficial for the patients in terms of less pain and rapid recovery with similar oncological outcomes. Therefore, we started a prospective study on laparoscopic surgery for early GBC in 2004. As this is the first prospective study for applying laparoscopy to malignant disease, we decided to plan the protocol to include only early GBC. Around 2010 was a time when many leading authors reported their initial experiences of advanced laparoscopy. Gumbs et al.[4] reported encouraging results of three patients who received laparoscopic extended cholecystectomy, with no morbidity or mortality. In 2010, we also reported our “initial experience of laparoscopic approach with suspected gallbladder cancer”[5]. Figure 1 shows our initial algorithm of patient care for suspected GBC. Endoscopic ultrasound was performed to determine liver invasion, and cases with liver invasion were treated with open radical cholecystectomy. In cases with peritoneal side tumors, intraoperative ultrasound was performed by experienced radiologists to rule out liver invasion. When there was no invasion, and the frozen section confirmed malignancy, laparoscopic extended cholecystectomy (which includes lymphadenectomy) was performed. Three trocars were used in the standard way for cholecystectomy. A thin layer of liver tissue was removed with the gallbladder to avoid bile spillage and to secure a safe margin. When frozen section confirmed malignancy, one or two trocars were additionally inserted for lymphadenectomy, and the pericholedochal, hilar, periportal, and common hepatic nodes were routinely dissected. Figure 2 shows the completion of lymphadenectomy.
After confirming the oncologic safety, laparoscopic surgery has been cautiously applied to GBC with liver invasion. To demonstrate this technique, we published a case report as a video article[6]. Laparoscopic cholecystectomy with en bloc resection of the liver bed was performed, followed by regional lymphadenectomy. Ultrasonic shears were used to dissect the superficial liver parenchyma, and Cavitron Ultrasonic Surgical Aspirator was used to dissect the deeper parenchyma. The report has shown that laparoscopic lymphadenectomy and liver resection can be safely performed. With encouraging advances in surgical technique, we can move forward to extended cholecystectomy with liver wedge resection.
The indication for laparoscopy was further expanded to operations including bile duct resection. A video article of extended cholecystectomy with bile duct resection was published[7]. The patient presented with postoperatively diagnosed GBC performed at another hospital. The cystic duct margin showed high grade dysplasia. Laparoscopic bile duct resection with lymph node dissection was performed. The bile duct was resected and retrocolic choledochojejunostomy was performed. The entire procedure of extended cholecystectomy, including lymphadenectomy, liver wedge resection, and bile duct resection, can be performed with laparoscopic procedure.
For technical tips, during laparoscopic extended cholecystectomy, a 3D flexible videoscope is usually used. This facilitates better orientation of the operative field, which makes equipment manipulation easy.
Additionally, when dividing small vessels, we prefer using a vessel sealing energy device rather than applying hemoclips. This technique shortens the operation time and can provide a cleaner operative field.
LONG-TERM OUTCOMES
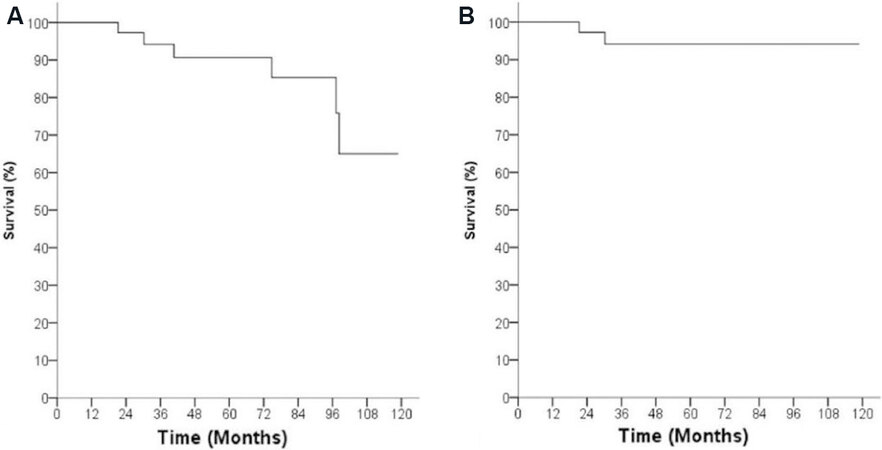
In 2015, we published our long-term outcomes after laparoscopic approach for early GBC[8]. During a ten-year period, 83 patients with suspected early GBC were enrolled in our prospective laparoscopic surgery protocol. Among these 83 patients, 45 patients had a pathologic diagnosis of GBC. The pathologic characteristics of the 45 patients are shown in Table 1. After a median follow-up period of 60 months for 45 patients, the overall survival rate was 90.7%, and the disease-specific 5-year survival rate was 94.2% [Figure 3]. There were no cases with local recurrence at the lymphadenectomy site or the gallbladder bed. From these results, we concluded that MIS for GBC is an oncologically safe operation.
Pathologic characteristics of patients with gallbladder cancer
| Variable | Data |
| T stage, n | |
| Tis | 2 |
| T1a | 10 |
| T1b | 8 |
| T2 | 25 |
| N stage, n | |
| Nx | 13 |
| N0 | 27 |
| N1 | 5 |
| No. of retrieved lymph nodes, | |
| Median (range) | 7 (1-15) |
| Tumor size, cm, median (range) | 3.2 (1.2-11.5) |
| Histologic differentiation, n | |
| Well differentiated | 29 |
| Moderately differentiated | 13 |
| Poorly differentiated | 3 |
| Angiolymphatic invasion | 8 |
| Perineural invasion | 4 |
| R status, R0 (%) | 45 (100) |
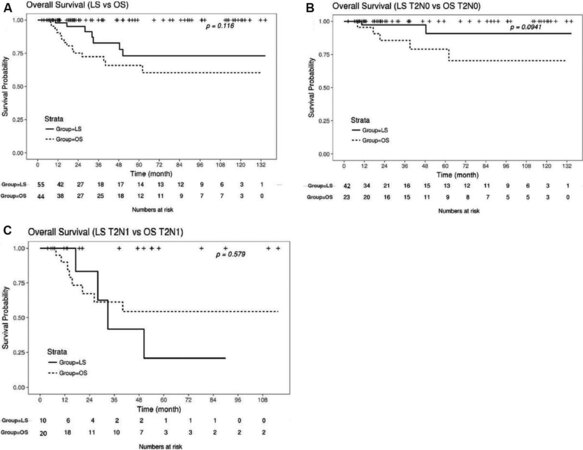
After accumulating 13 years of experience of laparoscopic extended cholecystectomy, we analyzed the oncologic outcomes of open vs. laparoscopic surgery for T2 GBC[9]. During the period of 2004 to 2017, 247 patients with GBC were treated were at our hospital. Among these patients, 151 patients had T2 cancer. After exclusion, a total of 99 patients were analyzed. The types of operations performed on the open surgery (OS) group and the laparoscopic surgery (LS) group are shown in Table 2. The OS group had more liver wedge resections than the LS group. The overall survival rates of the two groups are shown in Figure 4; there was no statistical difference between the two groups in overall survival rate. The entire group was subdivided into T2N0 group and T2N1 group to compare the overall survival according to nodal status. There was no significant difference between the OS group and LS group, in both the T2N0 subgroup and T2N1 subgroup. This outcomes show that laparoscopic surgery is compatible with open surgery even in T2 stage GBC.
Figure 4. (A) Overall survival rate of laparoscopic surgery (LS) group and open surgery (OS) group; (B) overall survival rate of T2N0 patients of OS group and LS group; and (C) overall survival rate of T2N1 patients of OS group and LS group.
Types of operations performed on the laparoscopic surgery and open surgery group
| LS group (n = 55) | OS group (n = 44) | |||||
| T2Nx | T2N0 | T2N1 | T2Nx | T2N0 | T2N1 | |
| (n = 3) | (n = 42) | (n = 10) | (n = 1) | (n = 23) | (n = 20) | |
| C + LND | 2 | 30 | 6 | 4 | 5 | |
| C + LND + LWR | 1 | 11 | 4 | 1 | 17 | 14 |
| C + LND + EHBDR | 1 | 2 | 1 | |||
For more advanced lesions, such as more than T3, further comparative studies are necessary to evaluate the oncologic safety of the laparoscopic approach.
LAPAROSCOPIC SURGERY FOR GALLBLADDER CANCER: AN EXPERT CONSENSUS STATEMENT
Despite these encouraging results, and an increasing number of reports on the feasibility of the laparoscopic approach for the treatment of GBC, there was no consensus among experts. In September 10th, 2016, a consensus meeting was held in Seoul, Korea, and the expert consensus statement on laparoscopic surgery for GBC was established[10]. Specific issues of this procedure were discussed among experts, such as concerns regarding laparoscopic surgery for GBC, application of laparoscopic surgery for GBC, laparoscopic extended cholecystectomy for GBC, and laparoscopic reoperation for postoperatively diagnosed GBC. The experts concluded that laparoscopic surgery does not worsen the prognosis of patients with early stage GBC, and that the postoperative and survival outcomes of highly selected patients were favorable.
Before this meeting was held, an international survey was undertaken of expert surgeons in the field of GBC surgery, and the results were published along with a review of the literature on the outcomes of laparoscopic surgery for GBC[11]. The majority of surgeons agreed that laparoscopic surgery has an acceptable role for suspicious or early GBC, and that laparoscopic extended cholecystectomy has a value comparable to that of open surgery in selected patients with GBC. But the selection criteria for laparoscopic surgery for overt GBC, and the detailed techniques varied among surgeons.
The results of perioperative outcomes and survival outcomes in this review are shown in Tables 3 and 4, respectively.
Perioperative outcomes of published case series in which more than 5 patients with gallbladder cancer underwent laparoscopic extended cholecystectomy
| Publication | Number of GBC patients | Indication | Open conversion (reason) | Operative time, min | Blood loss, mL | Complication, n (%) | Hospital stay, days |
| Cho et al.[5] | 18 | Primary | 1 (portal vein injury) | 190* | 50* | 3 (16.7) | 4* |
| de Aretxabala et al.[12] | 7 | Completion | 2 (LN metastasis, bile duct injury) | NA | NA | 0 | 3 |
| Gumbs et al.[13] | 15 | Primary (10), completion (5) | 1 (CBD resection) | 220 | 160 | 0 | 4 |
| Agarwal et al.[14] | 24 | Primary (20), completion (4) | 0 | 270* | 200* | 3 (12.5) | 5* |
| Itano et al.[15] | 16 | Primary (16) | 0 | 360 | 152 | 1 (5.2) | 9 |
| Shirobe et al.[16] | 11 | Primary (4), completion (7) | 1 (CBD resection) | 196 | 92 | 1 (9.1) | 6 |
| Yoon et al.[8] | 30 | Primary | 1 (portal vein injury) | 205* | 100* | 6 (18.8) | 4* |
| Palanisamy et al.[17] | 1 | Primary | 0 | 213 | 196 | 4 (28.6) | 5 |
Oncologic outcomes of published case series that included more than 5 patients with gallbladder cancer who underwent laparoscopic extended cholecystectomy
| Publication | 7th AJCC stage | Curative resection, % | No. of retrieved LNs | Recurrence (local/systemic) | Survival |
| Cho et al.[5] | I (6), II (8), IIIB (2) | 100 | 8* | 0 | NA |
| de Aretxabala et al.[12] | NA | NA | 6 | 1 (systemic) | NA |
| Gumbs et al.[13] | I (4), II (8), IIIB (3) | 100 | 4 | 2 (local, systemic) | NA |
| Agarwal et al.[14] | I (3), II (10), IIIA (6), IIIB (5) | 100 | 10* | 1 (local) | NA |
| Itano et al.[15] | I (3), II (13) | 100 | 13 | 0 | NA |
| Shirobe et al.[16] | I (3), II (6), IIIB (2) | 82 | 13 | 2 (local + systemic, local) | 5-year survival rate: 100% for T1b 83.3% for T2 |
| Yoon et al.[8] | I (8), II (17), IIIB (5) | 100 | 7* | 4 (systemic) | 5-year survival rate: 94.2% |
| Palanisamy et al.[17] | II (8), IIIA (1), IIIB (3) | 100 | 8* | 2 (systemic) | 5-year survival rate; 68.75% |
PERSPECTIVES OF MIS INCLUDING ROBOTIC SURGERY
The development of minimally invasive surgery in the field of hepato-pancreato-biliary surgery was truly remarkable. A collective effort of surgeons has led to a wide dissemination of advanced laparoscopy in the hepato-pancreato-biliary field. Not only are the experts at high volume centers performing these high-end operations, but many surgeons around the globe now routinely perform laparoscopic hepato-pancreato-biliary surgery.
The advent of robotic surgery was another milestone in the history of surgery. Although many surgeons have readily acknowledged this technique, the benefits are still a matter of debate. Advocates maintain that robotic surgery is superior, due to the fine and precise movements and magnified 3D vision. Others are concerned about the loss of tactile feedback, limited array of instruments, and the cost issue. But even with this debate, many surgeons have already reported huge experiences in robotic
When performing robotic pancreaticoduodenectomy, there is a definite benefit in facilitating anastomosis with higher degree of freedom. When pancreaticojejunostomy is performed by laparoscopy, there is limited freedom of instrument motion, and the needle holder manipulation is difficult. In contrast, when pancreaticojejunostomy is performed by the robot-assisted method, suturing can be performed almost the same as in open surgery, without any limitation of movement.
There have been several early reports of robotic surgery for GBC[3,18,19]. In 2020, Belli et al.[20] reported their experience on robotic surgery for 8 patients with GBC, with a mean operative time of 147 minutes and a 0% conversion rate. However, some issues need to be addressed. Surgery of GBC ranges from relatively simple cholecystectomy and lymphadenectomy to liver resection and bile duct resection. Early GBC may only require cholecystectomy with or without lymphadenectomy. Advanced cases may require extensive surgery with liver resection, and/or bile duct resection. If the operation is complicated, either laparoscopy and robot-assisted surgery can be chosen depending on each surgeon’s preference. But in relatively simple surgery, the issue of cost-effectiveness matters.
Due to the monopoly of Da Vinci robotics, the cost of robotic surgery is still high. Many patients in Korea do not have insurance coverages for robotic surgery, forcing them to pay the high cost of robotic surgery out-of-pocket. For pancreas and liver surgery, the operative type is planned preoperatively. In contrast, in the treatment of GBC, the decision to proceed with a radical operation is often decided according to the results of the intraoperative frozen section pathology. Therefore, routine use of robots for any stage of GBC can be too expensive. Another demerit of robot-assisted surgery is the lack of proper instruments. When liver resection is required, parenchymal transection may be difficult, as there is no cavitron ultrasonic surgical aspirator in robotic surgery. Harmonic scalpel is frequently used as well for parenchymal transection in liver surgery. However, a robotic harmonic scalpel with endo-wrist movement has yet to be developed, diminishing the advantage of robotic surgery. In many cases of GBC, lymphadenectomy can be the only necessary procedure for an extended cholecystectomy. This procedure can be performed superbly with laparoscopic surgery. The benefits of choosing the robotic system for just the lymphadenectomy are questionable.
The dissemination of robotic surgery may be different from the dissemination of laparoscopic surgery. When laparoscopic surgery was first introduced, there was the very definite, obvious benefit of reduced scars and faster recovery compared to open surgery. The only concern was to ensure the oncologic safety. However, robotic surgery has no obvious benefits over laparoscopic surgery, which makes adoption of the procedure still a matter of debate, even decades after the introduction of robotic surgery.
Breakthrough innovations in the field of surgery are constantly happening. We are currently debating the pros and cons of laparoscopic and robotic surgery, but as is outlined in this editorial by professor
CONCLUSION
Laparoscopic surgery is a safe, effective alternative for open surgery in the treatment of gallbladder cancer. The benefits of robotic surgery should be proven with further research.
DECLARATIONS
Authors’ contributionsManuscript drafting: Lee JS
Made substantial contributions to conception and design of the study, editing, and administrative support: Han HS
Provided administrative, technical, and material support: Yoon YS, Cho JY, Lee HW, Lee BR, Kim MW,
Not applicable.
Financial support and sponsorshipNone.
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Author(s) 2021.
REFERENCES
1. Burasakarn P, Thienhiran A, Hongjinda S, Fuengfoo P. The optimal extent of surgery in T2 gallbladder cancer and the need for hepatectomy: a meta-analysis. Asian J Surg 2021; doi: 10.1016/j.asjsur.2021.05.005.
2. Agarwal AK, Kalayarasan R, Javed A, Gupta N, Nag HH. The role of staging laparoscopy in primary gall bladder cancer--an analysis of 409 patients: a prospective study to evaluate the role of staging laparoscopy in the management of gallbladder cancer. Ann Surg 2013;258:318-23.
3. Byun Y, Choi YJ, Kang JS, et al. Robotic extended cholecystectomy in gallbladder cancer. Surg Endosc 2020;34:3256-61.
4. Gumbs AA, Hoffman JP. Laparoscopic completion radical cholecystectomy for T2 gallbladder cancer. Surg Endosc 2010;24:3221-3.
5. Cho JY, Han HS, Yoon YS, Ahn KS, Kim YH, Lee KH. Laparoscopic approach for suspected early-stage gallbladder carcinoma. Arch Surg 2010;145:128-33.
6. Kim S, Yoon YS, Han HS, Cho JY, Choi Y. Laparoscopic extended cholecystectomy for T3 gallbladder cancer. Surg Endosc 2018;32:2984-5.
7. Han S, Yoon YS, Han HS, Lee JS. Laparoscopic bile duct resection with lymph node dissection for gallbladder cancer diagnosed after laparoscopic cholecystectomy. Surg Oncol 2020;35:475.
8. Yoon YS, Han HS, Cho JY, et al. Is laparoscopy contraindicated for gallbladder cancer? J Am Coll Surg 2015;221:847-53.
9. Jang JY, Han HS, Yoon YS, Cho JY, Choi Y. Retrospective comparison of outcomes of laparoscopic and open surgery for T2 gallbladder cancer - thirteen-year experience. Surg Oncol 2019;29:142-7.
10. Han HS, Yoon YS, Agarwal AK, et al. Laparoscopic surgery for gallbladder cancer: an expert consensus statement. Dig Surg 2019;36:1-6.
11. Yoon YS, Han HS, Agarwal A, et al. Survey results of the expert meeting on laparoscopic surgery for gallbladder cancer and a review of relevant literature. Dig Surg 2019;36:7-12.
12. Aretxabala X, Leon J, Hepp J, Maluenda F, Roa I. Gallbladder cancer: role of laparoscopy in the management of potentially resectable tumors. Surg Endosc 2010;24:2192-6.
13. Gumbs AA, Jarufe N, Gayet B. Minimally invasive approaches to extrapancreatic cholangiocarcinoma. Surg Endosc 2013;27:406-14.
14. Agarwal AK, Javed A, Kalayarasan R, Sakhuja P. Minimally invasive versus the conventional open surgical approach of a radical cholecystectomy for gallbladder cancer: a retrospective comparative study. HPB (Oxford) 2015;17:536-41.
15. Itano O, Oshima G, Minagawa T, et al. Novel strategy for laparoscopic treatment of pT2 gallbladder carcinoma. Surg Endosc 2015;29:3600-7.
16. Shirobe T, Maruyama S. Laparoscopic radical cholecystectomy with lymph node dissection for gallbladder carcinoma. Surg Endosc 2015;29:2244-50.
17. Palanisamy S, Patel N, Sabnis S, et al. Laparoscopic radical cholecystectomy for suspected early gall bladder carcinoma: thinking beyond convention. Surg Endosc 2016;30:2442-8.
18. Araujo RLC, de Sanctis MA, Coelho TRV, Felippe FEC, Burgardt D, Wohnrath DR. Robotic surgery as an alternative approach for reoperation of incidental gallbladder cancer. J Gastrointest Cancer 2020;51:332-4.
19. Goel M, Khobragade K, Patkar S, Kanetkar A, Kurunkar S. Robotic surgery for gallbladder cancer: operative technique and early outcomes. J Surg Oncol 2019;119:958-63.
20. Belli A, Patrone R, Albino V, et al. Robotic surgery of gallbladder cancer. Mini-invasive Surg 2020;4:77.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Lee JS, Han HS, Yoon YS, Cho JY, Lee HW, Lee B, Kim M, Jo Y. Minimally invasive surgery for gallbladder cancer at an expert center. Mini-invasive Surg 2021;5:57. http://dx.doi.org/10.20517/2574-1225.2021.139
AMA Style
Lee JS, Han HS, Yoon YS, Cho JY, Lee HW, Lee B, Kim M, Jo Y. Minimally invasive surgery for gallbladder cancer at an expert center. Mini-invasive Surgery. 2021; 5: 57. http://dx.doi.org/10.20517/2574-1225.2021.139
Chicago/Turabian Style
Lee, Jun-Suh, Ho-Seong Han, Yoo-Seok Yoon, Jai-Young Cho, Hae-Won Lee, Boram Lee, Moonhwan Kim, Yeongsoo Jo. 2021. "Minimally invasive surgery for gallbladder cancer at an expert center" Mini-invasive Surgery. 5: 57. http://dx.doi.org/10.20517/2574-1225.2021.139
ACS Style
Lee, J.S.; Han H.S.; Yoon Y.S.; Cho J.Y.; Lee H.W.; Lee B.; Kim M.; Jo Y. Minimally invasive surgery for gallbladder cancer at an expert center. Mini-invasive. Surg. 2021, 5, 57. http://dx.doi.org/10.20517/2574-1225.2021.139
About This Article
Special Issue
Copyright
Data & Comments
Data
 Cite This Article 17 clicks
Cite This Article 17 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.