Computer-aided quality assessment (CAQ): the next step for artificial intelligence in colonoscopy?
Abstract
Recent developments and breakthroughs in artificial intelligence (AI) for colonoscopy have the potential to improve the quality of colonoscopy. Computer-aided detection for colorectal polyps has been shown to increase the adenoma detection rate by more than 10%. Furthermore, recently developed computer-aided quality assessment (CAQ) systems, such as real-time withdrawal speed monitoring, are expected to provide additional gain in colonoscopy practice. However, the added clinical value of combining AI techniques is uncertain. This paper provides an overview of the latest evidence on CAQ systems and identifies knowledge gaps that need to be filled before widespread implementation.
Keywords
INTRODUCTION
Colorectal cancer (CRC) is the third most common cancer and the second leading cause of cancer death worldwide[1]. Colonoscopy screening with detection and removal of neoplastic, precancerous polyps such as adenomas is performed in many countries for the prevention and early detection of CRC[2,3]. The effectiveness of colonoscopy screening depends on a high adenoma detection rate (ADR), which is the proportion of colonoscopies where at least one adenoma is detected[4].
Cause of missing adenoma
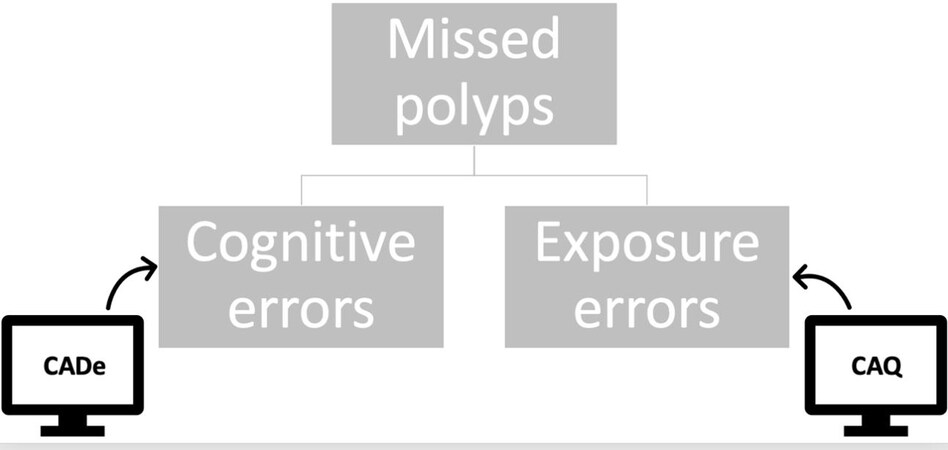
Although detection of adenomas is key for cancer prevention, almost one-fourth of adenomas are missed at CRC screening. Missed adenomas at screening are a main reason for CRC after screening with colonoscopy, so-called interval cancer[5,6], of which 89% are deemed avoidable[7]. Missed adenomas can be either the result of recognition failures, such as when endoscopists fail to recognize adenomas that are visualized on the screen (cognitive errors), or due to blind spots and incomplete mucosal exposure that is related to the speed during colonoscopy withdrawal, endoscopists’ skill, degree of bowel cleansing, and other factors (exposure errors)[5,7,8].
Computer-aided detection
Artificial intelligence (AI) by computer-aided detection (CADe) aims at improving ADR by reducing cognitive errors of endoscopists and thus reducing the rate of missed polyps[9,10]. Since ADR is known to be inversely associated with the risk of interval CRC[11], there is hope that AI reduces cancer incidence after colonoscopy. However, clinical trial evidence is currently lacking to support this claim.
CADe systems based on deep learning identifiy polyps in real time and display visual alerts on the monitor for the endoscopist. CADe systems showed an increase in ADR of 10% compared to colonoscopies done without CADe[9,10]. A recent randomized tandem study found that the adenoma miss rate with CADe was significantly lower compared to standard colonoscopy (13.89% vs. 40.00%, P < 0.0001)[12]. Furthermore, CADe does not reduce the miss rate of advanced adenomas[12], those with the highest immediate impact on colorectal cancer risk. The incremental benefit of CADe on CRC incidence therefore remains to be investigated.
COMPUTER-AIDED QUALITY ASSESSMENT SYSTEM
Since CADe only addresses cognitive errors, recent developments in computer-aided techniques have focused on measures to overcome exposure errors [Figure 1]. These tools can be defined as computer-aided quality assessment (CAQ) systems and is a third group of computer-aided techniques in colonoscopy along with CADe and computer-aided diagnosis. CAQ is expected to increase mucosal exposure during colonoscopy and promote more careful visualization. Specific measures of CAQ include the withdrawal speed monitoring and blind spot detector, which warn the endoscopist whenever the speed limit has been exceeded and there is a blind spot during withdrawal of endoscopes, respectively[13-15].
Figure 1. Missed polyps are caused by either cognitive errors or exposure errors. CADe: Computer-aided detection; CAQ: computer-aided quality assessment.
Withdrawal time is considered a surrogate quality indicator of mucosal inspection, influencing ADR. A withdrawal time of at least 6 min is recommended and may indicate a more careful inspection of the colorectal mucosa during screening colonoscopy[16-18]. However, the recommended withdrawal time is sometimes ignored due to the busyness of clinical practice, the lack of knowledge on guidelines, or other reasons.
Apart from withdrawal time, it is also important to maintain a stable maneuvering and uniform speed throughout withdrawal. This is especially crucial when passing colonic flexures where the endoscopist can experience endoscope slipping, resulting in quickly changing frames and blurry images, reduced mucosal exposure, and increased blind spots. Therefore, identifying withdrawal speed and identifying blind spots during colonoscopy are considered two major goals of the CAQ systems. CAQ is especially expected to help less experienced endoscopists achieve a better ADR in this regard.
Added clinical value of CADe and CAQ
As described above, several randomized trials have shown that CADe during colonoscopy significantly increases ADR[9,10]. CAQ is also expected to improve ADR indirectly through slower, repetitive, and more conscious withdrawal. The added value of CADe combined with CAQ has recently been indicated in early studies from China investigating endoscopic AI-based monitoring of the withdrawal speed and blind spots and its efficacy to improve ADR [Table 1][13-15].
ADR comparison from studies investigating CADe and CAQ
Studies where CAQ has been applied and investigated
Assessing the combined use of CAQ and CADe allows an evaluation of the incremental effect of the two computer-aided techniques, which is starting to gain more attention. Two studies[13,14] investigated the combined use of CAQ and CADe and compared it to standard colonoscopy with no involvement of CAD techniques. They found that ADR was significantly improved with the combined use of CAQ and CADe during colonoscopy, compared to non-AI colonoscopy.
Another study[15] investigated the efficacy of CAQ and CADe, comparing the combined use of CAQ and CADe with standard, non-AI colonoscopy, as well as with CADe or CAQ alone. Both CADe alone and CAQ alone showed a significantly increased ADR compared to non-AI colonoscopy, but when compared to each other, there was no significant difference in ADR. CAQ combined with CADe compared to CADe alone yielded an ADR of 30.6% vs. 21.3%. CAQ combined with CADe compared to non-AI colonoscopy yielded an ADR of 30.6% vs. 14.8%. However, there was no significant difference in ADR between CAQ combined with CADe compared to CAQ alone [Table 1]. Lastly, there was no significant difference in withdrawal time among any of the groups [Table 2].
Withdrawal time comparison from studies investigating CADe and CAQ
| Study | Withdrawal time in standard colonoscopy | Withdrawal time with CADe alone | Withdrawal time with CAQ alone | Withdrawal time with CAQ combined with CADe | P-value |
| Gong | 4.76 min | N/A | N/A | 6.38 min | |
| Su et al.(2020)[14] | 5.68 min | N/A | N/A | 7.03 min | |
| Yao et al.(2021)[15] | 9.71 min | 10.52 min | 10.14 min | 10.17 min |
^P = 0.056 *P = 0.302 **P = 0.413 |
Implementation of CAQ in colonoscopy: barriers and future
With only three studies, all performed in one country, investigating the added clinical value of applying CAQ alone and CAQ combined with CADe, there is a need for more studies such as these, especially from other countries, for external validation before implementation of CAQ can be recommended.
A concern with CAQ is the possibility that it can lead to an overload of information, especially when using two different monitors, and result in a negatively impacted ADR, longer withdrawal time without gains in ADR, or simply alert fatigue. Multiple alerts from more than one AI system during colonoscopy could exhaust endoscopists and reduce focus. These hurdles can be overcome by carefully finding the right sensitivity for alarm frequency and having all alerts on one monitor so that the endoscopist can focus on the same place rather than several.
CONCLUSION
CAQ combined with CADe is an effective method to further increase ADR during colonoscopy without prolonging the withdrawal time. Artificial intelligence could contribute to post hoc auditing of the colonoscopy quality or other steps within the whole logistic process of CRC screening. It is likely that these computer-aided techniques will be implemented in colonoscopy procedures in the near future.
DECLARATIONS
Authors’ contributionsMade substantial contributions to conception and design of the study and performed data analysis and interpretation; provided critical feedback and helped shape the research, analysis and manuscript: Barua I, Bretthauer M, Mori Y
Availability of data and materialsNot applicable.
Financial support and sponsorshipNone.
Conflicts of interestBarua I has received a travel grant from Olympus Norway. Mori Y has received consulting and speaking honorariums from Olympus Corp and has ownership interest in Cybernet Systems Corp. Bretthauer M has received consulting honorarium from Cybernet Systems Corp.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Author(s) 2022.
REFERENCES
1. Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2021;71:209-49.
2. Schreuders EH, Ruco A, Rabeneck L, et al. Colorectal cancer screening: a global overview of existing programmes. Gut 2015;64:1637-49.
3. Zauber AG, Winawer SJ, O’Brien MJ, et al. Colonoscopic polypectomy and long-term prevention of colorectal-cancer deaths. N Engl J Med 2012;23:687-96.
4. Kaminski MF, Regula J, Kraszewska E, et al. Quality Indicators for Colonoscopy and the Risk of Interval Cancer. N Engl J Med 2010;362:1795-803.
5. Zhao S, Wang S, Pan P, et al. Magnitude, risk factors, and factors associated with adenoma miss rate of tandem colonoscopy: a systematic review and meta-analysis. Gastroenterology 2019;156:1661-74.e11.
6. Robertson DJ, Lieberman DA, Winawer SJ, et al. Colorectal cancers soon after colonoscopy: a pooled multicohort analysis. Gut 2014;63:949-56.
7. Rex DK, Cutler CS, Lemmel GT, et al. Colonoscopic miss rates of adenomas determined by back-to-back colonoscopies. Gastroenterology 1997;112:24-8.
8. Anderson R, Burr NE, Valori R. Causes of post-colonoscopy colorectal cancers based on world endoscopy organization system of analysis. Gastroenterology 2020;158:1287-99.e2.
9. Barua I, Vinsard D, Jodal H, et al. Artificial intelligence for polyp detection during colonoscopy: a systematic review and meta-analysis. Endoscopy 2021;53:277-84.
10. Hassan C, Spadaccini M, Iannone A, et al. Performance of artificial intelligence in colonoscopy for adenoma and polyp detection: a systematic review and meta-analysis. Gastrointest Endosc 2021;93:77-85.e6.
11. Corley DA, Jensen CD, Marks AR, et al. Adenoma detection rate and risk of colorectal cancer and death. N Engl J Med 2014;370:1298-306.
12. Wang P, Liu P, Glissen Brown JR, et al. Lower adenoma miss rate of computer-aided detection-assisted colonoscopy vs routine white-light colonoscopy in a prospective tandem study. Gastroenterology 2020;159:1252-61.e5.
13. Gong D, Wu L, Zhang J, et al. Detection of colorectal adenomas with a real-time computer-aided system (ENDOANGEL): a randomised controlled study. Lancet Gastroenterol Hepatol 2020;5:352-61.
14. Su JR, Li Z, Shao XJ, et al. Impact of a real-time automatic quality control system on colorectal polyp and adenoma detection: a prospective randomized controlled study (with videos). Gastrointest Endosc 2020;91:415-24.e4.
15. Yao L, Zhang L, Liu J, et al. Effect of an artificial intelligence-based quality improvement system significantly improved the efficacy of computer-aided detection system in colonoscopy: a four-group parallel study. Endoscopy 2021:25.
16. Vavricka SR, Sulz MC, Degen L, et al. Monitoring colonoscopy withdrawal time significantly improves the adenoma detection rate and the performance of endoscopists. Endoscopy 2016;48:256-62.
17. Sawhney MS, Cury MS, Neeman N, et al. Effect of institution-wide policy of colonoscopy withdrawal time ≥ 7 minutes on polyp detection. Gastroenterology 2008;135:1892-8.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Barua I, Bretthauer M, Mori Y. Computer-aided quality assessment (CAQ): the next step for artificial intelligence in colonoscopy?. Mini-invasive Surg 2022;6:28. http://dx.doi.org/10.20517/2574-1225.2022.05
AMA Style
Barua I, Bretthauer M, Mori Y. Computer-aided quality assessment (CAQ): the next step for artificial intelligence in colonoscopy?. Mini-invasive Surgery. 2022; 6: 28. http://dx.doi.org/10.20517/2574-1225.2022.05
Chicago/Turabian Style
Barua, Ishita, Michael Bretthauer, Yuichi Mori. 2022. "Computer-aided quality assessment (CAQ): the next step for artificial intelligence in colonoscopy?" Mini-invasive Surgery. 6: 28. http://dx.doi.org/10.20517/2574-1225.2022.05
ACS Style
Barua, I.; Bretthauer M.; Mori Y. Computer-aided quality assessment (CAQ): the next step for artificial intelligence in colonoscopy?. Mini-invasive. Surg. 2022, 6, 28. http://dx.doi.org/10.20517/2574-1225.2022.05
About This Article
Copyright
Data & Comments
Data
 Cite This Article 13 clicks
Cite This Article 13 clicks











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.