A combined endoscopic and robotic approach for Mirizzi syndrome: the short- and long-term outcomes
Abstract
Aims: We present the technique of a combined endoscopic and robotic approach for Mirizzi syndrome (MS) and report the short- and long-term results.
Methods: Between July 2012 and August 2020, all patients with suspected MS underwent endoscopic retrograde cholangiopancreatography (ERCP) for diagnostic confirmation and placement of the biliary stent. Subtotal cholecystectomy was then performed with the assistance of a surgical robot. The common bile duct was closed with a cuff of the gallbladder over a biliary stent. ERCP was repeated 6-8 weeks after surgery to remove the biliary stent and confirm the patency of common bile duct. The operative outcomes and long-term results were prospectively collected.
Results: Twenty-two patients (10 males and 12 females) were included in the study. All patients underwent the planned robotic subtotal cholecystectomy and pre- and postoperative ERCP. The median age was 65.5 years (range 16-89 years). The median operative time was 212.5 min (range 125-510 min), and the median blood loss was 35 mL (range 7-700 mL). The median postoperative hospital stay was four days (range 3-15 days). Four patients (18.2%) developed postoperative complications including two intra-abdominal collections (9.1%), one wound infection (4.5%), and one atrial fibrillation with pneumonia (4.5%). The last patient also represented the only operative mortality. The median follow-up was 45.6 months; none developed recurrent cholangitis or jaundice, but one patient (4.5%) had a recurrent common bile duct stone which was successfully removed by ERCP.
Conclusion: The proposed combined endoscopic and robotic approach can provide favorable short- and long-term outcomes for patients with MS.
Keywords
INTRODUCTION
Mirizzi syndrome (MS) is a rare complication of gallstone disease[1,2]. Patients usually present with jaundice or acute cholangitis due to obstruction at the common hepatic duct by a gallstone impacted at the Hartmann’s pouch or the cystic duct. The main concern of MS is potential bile duct injury during cholecystectomy for the unaware[3]. Even if the preoperative diagnosis was correctly made, MS was previously considered a contraindication of minimally invasive approach due to anatomical distortion, dense adhesions, and intrinsic risk of bile duct injury[4,5].
To overcome the diagnostic and therapeutic challenges of MS, we previously reported our initial experience of the combined endoscopic and robotic approach for MS[6]. In brief, endoscopic retrograde cholangiopancreatography (ERCP) was performed to confirm the diagnosis of MS and for the placement of a biliary stent. Robotic subtotal cholecystectomy was then carried out with primary closure of the bile duct defect with a cuff of the gallbladder wall. ERCP was then repeated postoperatively, during which the biliary stent was removed, and patency and continuity of the bile duct were confirmed.
The short-term results of the first five patients who underwent the combined endoscopic/robotic approach were compared with those of the 17 historical cases who underwent the open or laparoscopic approach. The combined approach resulted in a lower conversion rate and shorter hospital stay. It was also associated with reduced blood loss and postoperative complications, although these results are not statistically significant[6]. However, the long-term outcomes of the combined approach were unknown despite favorable short-term results. Since our previous publication, we adopted the combined endoscopic/robotic approach in another 17 patients. Here, we report the short- and long-term outcomes of these 22 patients (including the previously reported five patients).
METHODS
Twenty-two consecutive patients with MS, between July 2012 and August 2020, who underwent the combined endoscopic and robotic approach for disease management, were included in the study. No patients with MS underwent an open or laparoscopic approach during the study period. Data were prospectively collected, which included patient demographics, clinical presentation, preoperative blood tests and imaging, intraoperative findings, and postoperative outcomes. Short-term outcomes included length of hospital stay and postoperative complications. Long-term outcomes included recurrent biliary stone, cholangitis, and survival. A retrospective analysis was performed on this dataset. The study was approved by the Joint Chinese University of Hong Kong-New Territories East Cluster Clinical Research Ethics Committee (CREC No. 2021.691).
Two classification systems were commonly used to describe the different types of MS: the McSherry classification and the Csendes classification. For the McSherry classification, Type I refers to external compression of the bile duct by the gallstone, while Type II refers to a fistula between the gallbladder and the common bile duct[7]. This study uses the Csendes classification, described as follows: Type I, extrinsic compression of the bile duct; Type II, cholecystocholedochal fistula involving less than 1/3 of bile duct circumference; Type III, fistula involving up to 2/3 of a bile duct; Type IV, fistula with the complete destruction of bile duct wall; and Type V, presence of cholecystoenteric fistula together with any other type of MS[8,9].
Preoperative imaging and preparation
Ultrasound (US) was the initial investigation routinely used for biliary disease. For patients with suspected MS, computed tomography (CT) or magnetic resonance imaging (MRI) was also performed. In addition to delineating biliary anatomy, cross-sectional imaging was helpful in excluding gallbladder cancer or cholangiocarcinoma, which might give rise to a similar clinical picture to MS[10]. ERCP was performed for suspected or confirmed MS, as not only did it help confirm the diagnosis of MS, but it also enabled clearance of any concomitant common duct stone and placement of a plastic stent[10]. This restored bile flow and allowed cholangitis to subside before definitive surgical treatment. The operation was then performed once liver function normalized and sepsis subsided.
Operative technique
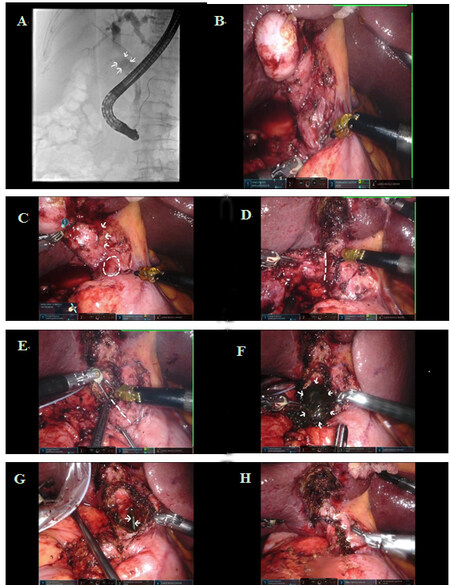
The operation was performed with the da Vinci S robotic system (Intuitive Surgical, Sunnyvale, CA, USA), which was upgraded to the da Vinci Xi in December 2017. Four robotic ports were placed for the camera and robotic instruments. Additional 5 and 10 mm assistant ports were inserted for retraction, suction, and passage of suture. The intended operation was a subtotal cholecystectomy. The aim of the operation was to remove most of the gallbladder, leaving a cuff of tissue near the common duct for primary closure [Figure 1]. The details of the operation technique have been reported previously[6].
Figure 1. (A) Endoscopic retrograde cholangiopancreatography (ERCP) showing stone (arrows) located at cholecystocholedochal fistula. (B-H) Operative views showing robotic subtotal cholecystectomy for Mirizzi syndrome: (B) outlook of the Calot’s triangle after omental adhesion taken down; (C) adherent Calot’s triangle (arrows), where the culprit stone was located by operative ultrasound (broken circle); (D) gallbladder dissected from liver with fundus first approach (the dotted line indicates the line for subtotal cholecystectomy); (E) the dotted line indicates the line for incision over Hartmann’s pouch for stone retrieval; (F) the culprit stone (arrows) to be removed; (G) biliary stent (arrows) as seen through cholecystocholedochal fistula; and (H) gallbladder cuff closed over the bile duct with a single layer continuous suture (arrows).
Postoperative management
Patients were discharged once they were fully mobilized and had adequate oral intake. They were readmitted 4-6 weeks postoperatively for repeat ERCP as a day procedure for stent removal and final check on bile duct patency. Patients were followed up every three months in the first year after the operation and then every six months afterward with liver function tests and complete blood counts before each follow-up. US was arranged six months after the operation and then as required afterward. CT or MRI was performed in the case of a suspicious US finding. Readmission for acute cholangitis, evidence of recurrent biliary stone on follow-up imaging (US, CT, or MRI), mortality, and cause of death were recorded during patient follow-up.
Outcome measurement
Operative complications were graded according to the Clavien-Dindo classification[11]. Operative mortality was defined as death within 90 days after operation. Continuous variables were expressed as median (range) and categorical variables were expressed as number (%).
RESULT
There were 10 male and 12 female patients in this study. All patients had the correct preoperative diagnosis of MS and underwent the planned robotic subtotal cholecystectomy and pre- and postoperative ERCP. The patient demographics and disease characteristics are shown in Table 1. The median age was 65.5 years (range 16-89 years). Thirteen patients presented with obstructive jaundice, while nine patients presented with acute cholangitis, with median serum bilirubin at 113 µmol/L (range 34-389 µmol/L). All patients except one had US as the initial imaging modality, and all patients had either CT or MRI for definitive diagnosis. The distribution of MS types according to Csendes’ Classification was as follows: Type I, 5 (22.7%); Type II, 11 (50%); Type III, 0 (0%); Type IV, 1 (4.5%); and Type V, 5 (22.7%). The median size of stone was 1.5 cm (range 1-3 cm) [Table 1].
Patient demographics and disease characteristics
| Patient no. | Age | Sex | Type of Mirizzi syndrome | Stone size (cm) | Presentation | Bilirubin on presentation (µmol/L) | Pre-operative imaging |
| 1 | 80 | F | II | 1.5 | Cholangitis | 97 | US, CT |
| 2 | 52 | F | II | 1.2 | Jaundice | 182 | US, CT |
| 3 | 67 | F | V | 2.6 | Cholangitis | 102 | US, CT, MRI |
| 4 | 74 | F | I | 2 | Jaundice | 255 | US, CT |
| 5 | 46 | M | I | 1.2 | Jaundice | 57 | US, MRI |
| 6 | 70 | F | II | 1.5 | Jaundice | 95 | US, CT, EUS |
| 7 | 41 | F | II | 1.2 | Cholangitis | 105 | US, CT |
| 8 | 56 | F | II | 2 | Cholangitis (MOF) | 85 | US, CT |
| 9 | 34 | F | II | 1.5 | Jaundice | 171 | US, CT |
| 10 | 16 | M | I | 1.3 | Jaundice | 158 | US, CT, MRI |
| 11 | 59 | M | V | 2 | Cholangitis | 219 | US, CT |
| 12 | 56 | M | V | 1 | Jaundice | 125 | US, CT, MRI |
| 13 | 68 | F | V | 1.8 | Cholangitis | 43 | US, CT |
| 14 | 67 | F | II | 2 | Cholangitis | 189 | US, CT |
| 15 | 56 | M | II | 1.5 | Jaundice | 141 | US, CT |
| 16 | 88 | M | V | 1.5 | Jaundice | 127 | US, CT |
| 17 | 56 | M | I | 1.5 | Jaundice | 389 | US, CT |
| 18 | 64 | M | II | 1 | Cholangitis | 121 | US, CT |
| 19 | 76 | M | II | 1.5 | Cholangitis | 85 | US, CT |
| 20 | 80 | M | IV | 3 | Jaundice | 34 | US, PET CT |
| 21 | 89 | F | I | 1.5 | Jaundice | 80 | US, CT |
| 22 | 73 | F | II | 2.5 | Jaundice | 96 | CT |
All patients had successful ERCP with deep cannulation and stent insertion to the common bile duct before their operation. The operative outcomes are shown in Table 2. The median operative time was 212.5 min (range 125-510 min) and the median blood loss was 35 mL (range 7-700 mL). One patient needed a perioperative transfusion. The median postoperative hospital stay was four days (range 3-15 days). Four patients (18.2%) developed postoperative complications (one Grade I, one Grade II, one Grade IIIa, and one Grade V), two patients had intra-abdominal collections (one required percutaneous drainage), one patient had wound infection, and one patient had atrial fibrillation and pneumonia.
Perioperative and long-term outcomes
| Patient no. | Additional procedure | Choledo-choscopy | Operative time (min) | Blood loss (ml) | Post-operative hospital stay (days) | Mortality | Morbidity | Follow-up period (months) | Recurrent stone | Recurrent cholangitis |
| 1 | No | Yes | 325 | 150 | 5 | No | No | 100.4 | No | No |
| 2 | No | No | 243 | 20 | 4 | No | No | 85.0 | No | No |
| 3 | DF | Yes | 510 | 100 | 4 | No | No | 92.0 | No | No |
| 4 | No | Yes | 273 | 300 | 4 | No | No | 89.0 | No | No |
| 5 | No | No | 215 | 20 | 3 | No | No | 87.4 | No | No |
| 6 | No | No | 180 | 5 | 4 | No | No | 59.5 | No | No |
| 7 | No | No | 160 | 10 | 4 | No | No | 64.1 | No | No |
| 8 | No | No | 150 | 20 | 3 | No | No | 17.0 | No | No |
| 9 | No | No | 136 | 10 | 4 | No | No | 35.0 | No | No |
| 10 | No | Yes | 220 | 20 | 6 | No | Wound infection | 9.7 | No | No |
| 11 | DF | No | 220 | 20 | 10 | No | Intra-abdominal collection | 68.0 | No | No |
| 12 | DF | No | 240 | 150 | 6 | No | No | 60.7 | No | No |
| 13 | DF+CF | Yes | 207 | 20 | 5 | No | No | 54.3 | No | No |
| 14 | Deroofing of liver cyst | No | 210 | 700 | 11 | No | No | 21.7 | No | No |
| 15 | No | No | 137 | 50 | 4 | No | No | 45.6 | No | No |
| 16 | DF+CF+GJ+T-tube | Yes | 500 | 350 | 15 | Yes | AF, pneumonia | NA | NA | NA |
| 17 | No | No | 127 | 5 | 4 | No | No | 2.7 | No | No |
| 18 | IOC | Yes | 310 | 50 | 4 | No | No | 29.0 | No | No |
| 19 | No | No | 157 | 200 | 6 | No | No | 28.6 | No | No |
| 20 | No | Yes | 319 | 100 | 6 | No | Intra-abdominal collection | 25.7 | Yes | No |
| 21 | No | No | 155 | 50 | 6 | No | No | 9.9 | No | No |
| 22 | No | No | 125 | 10 | 4 | No | No | 6.4 | No | No |
The last patient (Patient 16) represented the only operative mortality in this series (mortality rate of 4.5%). This patient was an 88-year-old gentleman who developed duodenal obstruction over time after initial ERCP and stent insertion. In addition to the offending stone causing MS, a large distal common bile duct stone was also noted at ERCP, and the decision was taken for its retrieval during surgery. Robotic subtotal cholecystectomy, takedown of cholecystoduodenal and cholecystocolic fistulae, exploration of the common bile duct, and gastrojejunostomy were performed. This was also the only patient in the series in whom the biliary stent was removed and a T-tube was placed, as postoperative ERCP was deemed not feasible due to duodenal obstruction. Gallbladder tissue was sent for frozen section during surgery due to concerns of malignancy, but it returned as inflammation only. The duration of the operation was long due to additional procedures (repair of cholecystoenteric fistulae, choledochoscopy, and gastrojejunostomy). Conversion to open was not considered, as slow but smooth progress was made, and the patient’s condition remained stable throughout the operation. The patient developed atrial fibrillation and pneumonia after surgery, but there was no surgical complication. The final pathology of the gallbladder specimen revealed adenocarcinoma, which was also the only malignancy found in this series. He was discharged to convalescence hospital on Postoperative Day 15 but was admitted one day later with fever and desaturation. He succumbed on Day 35 due to septic shock and desaturation secondary to a severe chest infection.
Five patients (22.7%) were found to have residual common bile duct stones on postoperative ERCP; all stones were successfully removed together with the biliary stent. The median period of follow-up was 45.6 months (range 2.7-100.4 months). The patient with the shortest follow-up was lost to follow-up after the postoperative ERCP. One patient (Patient 14) died 21 months after surgery. She had underlying polycystic liver and kidney disease complicated by tuberculosis infection and empyema thoracis. Her cause of death was liver and renal failure, without any evidence of recurrent biliary stone or cholangitis. No other patient developed recurrent cholangitis or jaundice, but one patient (4.5%) was found to have recurrent biliary stone on follow-up imaging. The recurrent stone was a common bile duct stone that was successfully removed by ERCP, and the patient remained well afterward.
DISCUSSION
MS is a rare disease that can be difficult to diagnose and manage. A high index of suspicion and the liberal use of CT or MRI can help establish the diagnosis. We strongly recommend ERCP for any patients suspected to have MS, both for diagnostic confirmation and for placement of a biliary stent to provide a road map for subsequent operation. Further, it is not unusual for patients with MS to present with jaundice and sepsis; stenting can therefore relieve biliary obstruction and improve liver function before surgery. Although ERCP can provide a correct diagnosis of MS, it is difficult to diagnose precise fistula extent of the bile duct wall preoperatively. Preoperative imaging such as US, CT, and MRI also cannot diagnose fistula involving less than 1/3, up to 2/3, or complete destruction of the bile duct wall. It means that a proper preoperative strategy is extremely difficult, and one should be cautious of higher-grade MS and additional findings such as cholecystoduodenal or cholecystocolic fistula during the operation. Nevertheless, our approach is feasible for all types of MS.
Historically, the minimally invasive approach was considered inappropriate in the management of MS, and it was only recommended for McSherry type I disease[12-14]. In a systemic review of the laparoscopic treatment of MS, 10 series including 124 patients between 1989 and 2008 were identified[5]. The overall conversion rate was 41%, with a reoperation rate of 6%, a complication rate of 20%, and a mortality rate of 0.8%. The median hospital stay was eight days (range 3-13 days). The review concluded that the operative outcomes of laparoscopic management of MS were unsatisfactory. A recent series including 11 patients undergoing laparoscopic management for McSherry type II MS reported more promising results. There was no conversion to open, the stone clearance rate was 100%, morbidity was 27.3%, and the median hospital stay was five days[15].
A combined endoscopic and open subtotal cholecystectomy was described for MS as early as 1999[16]. A combined endoscopic and laparoscopic approach was reported in 2000, but the conversion rate was still high at 22%[17]. A similar approach by another group in 2011 achieved a better conversion rate of 14%[18]. A recent series in 2016 from China reported 49 patients with McSherry type II MS undergoing the combined endoscopic and laparoscopic approach, and there were no cases of conversion[19]. When compared with a historical cohort of 57 patients who had open surgery for MS, the combined approach group had a significantly shorter hospital stay.
A combined endoscopic and robotic approach was reported by another center in the USA[20]. There were six patients, three of whom required a hepaticojejunostomy for biliary tract reconstruction. There was no open conversion or major complications. There was no readmission for recurrent stone or repeat biliary intervention. Our study demonstrated that the combined endoscopic and robotic approach was feasible and safe with no conversion; patients usually recovered rapidly from the operation with a median postoperative hospital stay of four days, low blood loss (median 35 mL) and an acceptable rate of complication (18.2%).
In our experience, robotic subtotal cholecystectomy can be safely performed for Types I-V disease, sparing patients from more major surgery including bile duct resection, hepaticojejunostomy, and jejunojejunostomy. The crux is to correctly identify the gallstone which is the culprit of the disease and its relation to the common bile duct. The use of laparoscopic US and indocyanine green (ICG) fluorescence are useful adjuncts. An incision is made on the gallbladder wall away from the bile duct, which is large enough to remove the stone as well as to examine the bile duct, confirmed by the presence of the biliary stent. Compared with the laparoscopic approach, the use of robotic instruments facilitates meticulous dissection and closure of any cholecystoenteric fistula by offering a steady operation platform. Closure of the cuff of gallbladder tissue over the bile duct is also enhanced by the articulated wristed robotic instruments. Although using a robot is more costly compared with laparoscopy, it can be more effective for higher-grade MS such as Types III-V, the dissection of which would benefit from robot-enhanced dexterity. The biliary stent also obviated the use of T-tube and its associated problems. Postoperative ERCP was used not only for stent removal but also for a final check on the status of the bile duct. In this series, up to 22.7% of patients had residual bile duct stones, all of which were successfully removed by ERCP.
Our combined endoscopic and robotic approach also resulted in satisfactory long-term outcomes. Only one patient (4.5%) was found to have recurrent common bile duct stone, which was successfully removed by ERCP. The low recurrent stone rate was in concordance with a recurrent choledocholithiasis rate of 4.55% in another study[19]. By preserving the common bile duct and avoiding hepaticojejunostomy at the index operation, subsequent access to the bile duct via ERCP is still feasible. All other patients remained symptom-free from recurrent biliary stone or cholangitis during follow-up. There was no occurrence of bile duct stricture. This confirmed that closure with a gallbladder cuff for the bile duct defect was safe and did not lead to subsequent stricture.
ICG is increasingly applied for biliary tract imaging during surgery. A recent randomized controlled trial has shown that fluorescence cholangiography was significantly superior to white-light imaging for visualization of most extrahepatic biliary structures before and after dissection[21]. We have applied ICG for biliary tract identification in a couple of cases of MS, and the initial results are encouraging but require further evaluation.
Peroral cholangioscopic lithotripsy is also an attractive option for the management of MS. It has been successfully applied for MS and cystic duct stones[22]. With further improvement of endoscopic instruments and techniques, MS may be effectively dealt with by endoscopic means alone in the future.
The current study represents the largest series of patients with MS who underwent a combined endoscopic and robotic approach for the disease. Given the rarity of the disease, we believe that the treatment outcomes provide evidence of the usefulness of this novel approach for MS. Although this combined approach was feasible for all types of MS, the majority of MS were Types I, II, or V in this study, and extrapolation of our study findings to Types III and IV MS needs to be further evaluated. In cases of high-grade MS, when our approach for primary closure of the bile duct is not feasible and bilio-enteric reconstruction is needed, the use of the robot can facilitate the bilio-enteric anastomosis. Further studies with larger patient numbers from different centers are required to support our study findings. In this study, all ERCPs were performed by hepatobiliary surgeons rather than gastroenterologists. The adoption of this combined approach in other centers will need active communication and collaboration between surgeons and gastroenterologists to ensure the objectives of the pre- and postoperative ERCPs are achieved. Lastly, a background of extensive training in hepatobiliary and minimally invasive surgery in order to achieve such desirable outcomes in this combined approach cannot be over-emphasized.
In conclusion, our proposed combined endoscopic and robotic approach can provide favorable short- and long-term outcomes for patients with MS. Currently, evidence for the standard of care in the management of MS is lacking; however, with the rapid improvement of technology and equipment, patients with MS should not be deprived of the advantages of minimally invasive surgery[23]. Further studies are indicated to confirm our findings.
DECLARATIONS
Authors’ contributionConception and design: Lee KF, Kung JWC
Data collection and analysis: Lee KF, Lo EYJ, Fung AKY
Manuscript drafting: Lee KF
Critical revision and final approval: Ng KKC, Wong J, Chong CCN, Lok HT, Fung AKY, Lo EYJ, Kung JWC
Availability of data and materialsData is stored in a database under the Department of Surgery, Prince of Wales Hospital, the Chinese University of Hong Kong.
Financial support and sponsorshipNone.
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateThe study was approved by the Joint Chinese University of Hong Kong-New Territories East Cluster Clinical Research Ethnics Committee (CREC No. 2021.691).
Consent for publicationWritten informed consent has been obtained from patient for publishing images and operative photos.
Copyright© The Author(s) 2022.
REFERENCES
1. Mirizzi PL. Sindrome del conducto hepatico. J Int Chir 1948;8:731-7.
2. Abou-Saif A, Al-Kawas FH. Complications of gallstone disease: Mirizzi syndrome, cholecystocholedochal fistula, and gallstone ileus. Am J Gastroenterol 2002;97:249-54.
3. Kok KY, Goh PY, Ngoi SS. Management of Mirizzi’s syndrome in the laparoscopic era. Surg Endosc 1998;12:1242-4.
4. Erben Y, Benavente-Chenhalls LA, Donohue JM, et al. Diagnosis and treatment of Mirizzi syndrome: 23-year Mayo Clinic experience. J Am Coll Surg 2011;213:114-9; discussion 120.
5. Antoniou SA, Antoniou GA, Makridis C. Laparoscopic treatment of Mirizzi syndrome: a systematic review. Surg Endosc 2010;24:33-9.
6. Lee KF, Chong CN, Ma KW, et al. A minimally invasive strategy for Mirizzi syndrome: the combined endoscopic and robotic approach. Surg Endosc 2014;28:2690-4.
7. McSherry CK, Ferstenberg H, Virshup M. The Mirizzi syndrome: suggested classification and surgical therapy. Surg Gastroenterol 1982;1:219-25.
8. Csendes A, Díaz JC, Burdiles P, Maluenda F, Nava O. Mirizzi syndrome and cholecystobiliary fistula: a unifying classification. Br J Surg 1989;76:1139-43.
9. Beltran MA, Csendes A, Cruces KS. The relationship of Mirizzi syndrome and cholecystoenteric fistula: validation of a modified classification. World J Surg 2008;32:2237-43.
10. Beltrán MA. Mirizzi syndrome: history, current knowledge and proposal of a simplified classification. World J Gastroenterol 2012;18:4639-50.
11. Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 2004;240:205-13.
12. Yeh CN, Jan YY, Chen MF. Laparoscopic treatment for Mirizzi syndrome. Surg Endosc 2003;17:1573-8.
13. Kwon AH, Inui H. Preoperative diagnosis and efficacy of laparoscopic procedures in the treatment of Mirizzi syndrome. J Am Coll Surg 2007;204:409-15.
14. Cui Y, Liu Y, Li Z, et al. Appraisal of diagnosis and surgical approach for Mirizzi syndrome. ANZ J Surg 2012;82:708-13.
15. Senra F, Navaratne L, Acosta A, Martínez-Isla A. Laparoscopic management of type II Mirizzi syndrome. Surg Endosc 2020;34:2303-12.
16. Hazzan D, Golijanin D, Reissman P, Adler SN, Shiloni E. Combined endoscopic and surgical management of Mirizzi syndrome. Surg Endosc 1999;13:618-20.
17. Chowbey PK, Sharma A, Mann V, et al. The management of Mirizzi syndrome in the laparoscopic era. Surg Laparosc Endosc Percutan Tech 2000;10:11-4.
18. Zheng M, Cai W, Qin M. Combined laparoscopic and endoscopic treatment for Mirizzi syndrome. Hepatogastroenterology 2011;58:1099-105.
19. Yuan H, Yuan T, Sun X, Zheng M. A minimally invasive strategy for Mirizzi syndrome type II: combined endoscopic with laparoscopic approach. Surg Laparosc Endosc Percutan Tech 2016;26:248-52.
20. Magge D, Steve J, Novak S, et al. Performing the difficult cholecystectomy using combined endoscopic and robotic techniques: how i do it. J Gastrointest Surg 2017;21:583-9.
21. Dip F, LoMenzo E, Sarotto L, et al. Randomized trial of near-infrared incisionless fluorescent cholangiography. Ann Surg 2019;270:992-9.
22. Bhandari S, Bathini R, Sharma A, Maydeo A. Usefulness of single-operator cholangioscopy-guided laser lithotripsy in patients with Mirizzi syndrome and cystic duct stones: experience at a tertiary care center. Gastrointest Endosc 2016;84:56-61.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Lee KF, Kung JWC, Lo EYJ, Fung AKY, Lok HT, Chong CCN, Wong J, Ng KKC. A combined endoscopic and robotic approach for Mirizzi syndrome: the short- and long-term outcomes. Mini-invasive Surg 2022;6:32. http://dx.doi.org/10.20517/2574-1225.2022.18
AMA Style
Lee KF, Kung JWC, Lo EYJ, Fung AKY, Lok HT, Chong CCN, Wong J, Ng KKC. A combined endoscopic and robotic approach for Mirizzi syndrome: the short- and long-term outcomes. Mini-invasive Surgery. 2022; 6(7): 32. http://dx.doi.org/10.20517/2574-1225.2022.18
Chicago/Turabian Style
Lee, Kit-Fai, Janet W. C. Kung, Eugene Y. J. Lo, Andrew K. Y. Fung, Hon-Ting Lok, Charing C. N. Chong, John Wong, Kelvin K. C. Ng. 2022. "A combined endoscopic and robotic approach for Mirizzi syndrome: the short- and long-term outcomes" Mini-invasive Surgery. 6, no.7: 32. http://dx.doi.org/10.20517/2574-1225.2022.18
ACS Style
Lee, K.F.; Kung JWC.; Lo EYJ.; Fung AKY.; Lok H.T.; Chong CCN.; Wong J.; Ng KKC. A combined endoscopic and robotic approach for Mirizzi syndrome: the short- and long-term outcomes. Mini-invasive. Surg. 2022, 6, 32. http://dx.doi.org/10.20517/2574-1225.2022.18
About This Article
Copyright
Data & Comments
Data

 Cite This Article 6 clicks
Cite This Article 6 clicks











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.