Novel surgical technique of robot-assisted thoracoscopic esophagectomy in the lateral decubitus position
Abstract
We performed robot-assisted thoracoscopic esophagectomy in lateral decubitus position (LDP) with camera rotation and manual hand control assignment to reproduce the visualization and manipulation of open esophagectomy or thoracoscopic esophagectomy in LDP. Four robotic ports and two 12 mm assistant ports were placed. The camera image for the operator was vertically and horizontally inverted by camera rotation to create an operative view similar to that achieved under open thoracotomy. We used a forward-oblique viewing endoscope with a 30° down-facing orientation. The mediastinal view was obtained by single lung ventilation, artificial pneumothorax by carbon dioxide insufflation, and trachea retraction by the assistant. The right and left hands were assigned to any combination of two out of the three arms depending on the situation. The remaining arm was used as an assistant to create an adequate surgical view. The robotic platform is useful to manage aortic injury by direct suturing.
Keywords
INTRODUCTION
Esophagectomy with regional lymphadenectomy plays a major role in esophageal cancer treatment. Thoracoscopic and laparoscopic minimally invasive esophagectomy procedures have been performed to reduce the surgical damage caused by esophageal surgery[1-3]. Recently, robot-assisted thoracoscopic esophagectomy (RATE) has become a widely adopted esophageal surgery. We introduced RATE in the lateral decubitus position (LDP) with the four-arm da Vinci Xi robotic system (Intuitive Surgical Inc., Sunnyvale, CA) as a curative operative procedure in 2018. The videoscopic images and hand control manipulation in the previously reported RATE in LDP[4-17] are the same as those in RATE in the prone position (PP)[18-24], where the arms for the right and left hands are introduced into the thoracic cavity from the cranial and caudal directions, respectively. We performed thoracoscopic esophagectomy (TE) in the LDP under the image rotated by 180°, where the left side was cranial and the upper side was ventral; this image is similar to the view under open thoracotomy[25]. We successfully reproduced the same endoscopic view and hand manipulation in RATE in the LDP as in our TE via inversion of the image with camera rotation and manual hand control assignment[26].
In this technical note, we present our surgical techniques of mediastinal dissection by RATE in LDP.
OPERATIVE PROCEDURE
As a radical surgical procedure for esophageal cancer, esophagectomy with three-field lymphatic dissection consisting of cervical, mediastinal, and abdominal dissections was performed. The operation was started from the thoracic procedure with RATE in LDP. After RATE, abdominal and supraclavicular cervical lymph node dissections were done simultaneously by two teams in the supine position. The abdominal lymphatic dissection was done by hand-assisted laparoscopic surgery. The gastric tube was selected as the primary reconstruction conduit and pulled up through the posterior mediastinum. The operation was finished with cervical anastomosis. In cases of non-curative resection, the retrosternal route was selected for elevation of the gastric tube to allow subsequent chemoradiotherapy of the residual tumor.
Preparation of RATE in LDP
For the thoracic procedure, patients were placed in the left LDP after intubation with a left-side
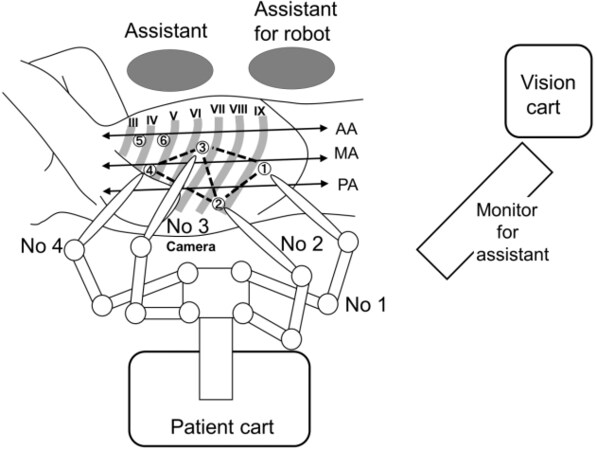
Figure 1. The trocar sites in the robot-assisted thoracoscopic esophagectomy in lateral decubitus position. Roman numerals show the rib numbers. Encircled numerals show the port sites. Ports 1-4 are the 8 mm da Vinci ports (Intuitive Surgical Inc.) connected to the corresponding robotic arms. Ports 5 and 6 are the 12 mm assistant ports. Port 6 is the AirSeal (ConMed) trocar. AA: Anterior axillary line; MA: middle axillary line; PA: posterior axillary line.
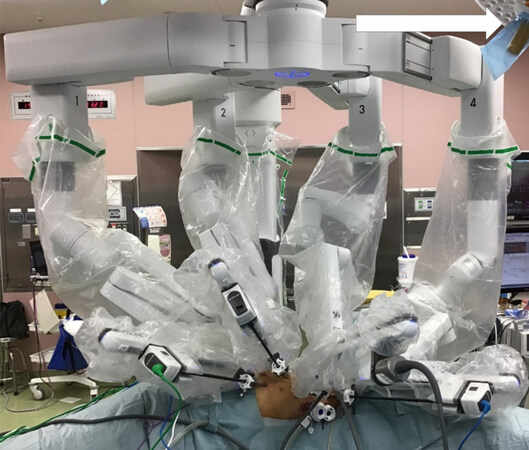
Figure 2. Final appearance after robot setup in robot-assisted thoracoscopic esophagectomy in lateral decubitus position. The patient cart is rolled in from the patient’s dorsal side and docked to the robotic ports. Arrows indicate the cranial direction.
Dissection of right recurrent laryngeal nerve nodes
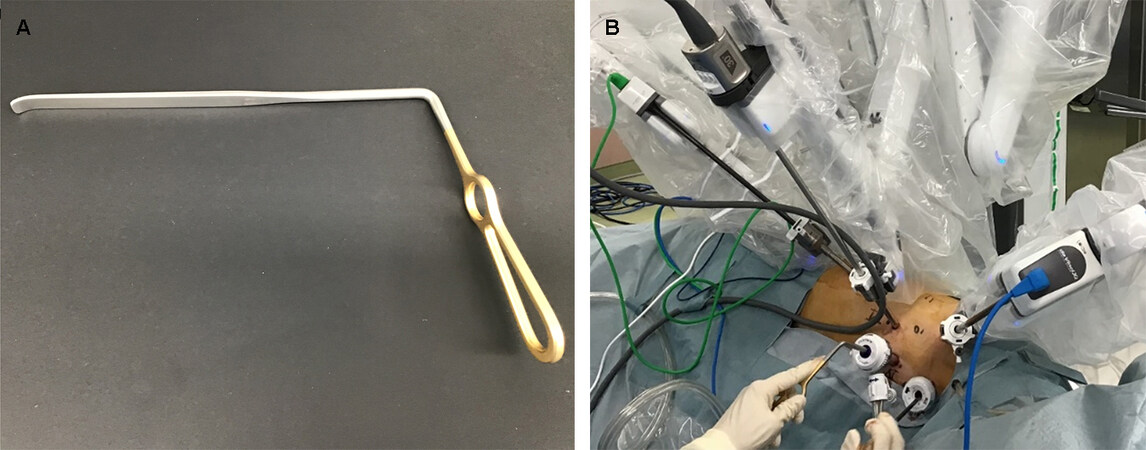
Parietal pleura on the upper mediastinum was incised along with the right vagal nerve and right subclavian artery. After exposing the right side of the tracheal wall, the lymph nodes around the right recurrent laryngeal nerve (RLN) were dissected while preserving the right RLN [Figure 4A]. After the identification of right RLN by its running route, the dividing point of right RLN from the vagal nerve was exposed. Fatty tissue containing lymph nodes was grasped by Cadiere forceps held by Arm 2 as assistant arm [Figure 4B]. The esophageal branches from RLN were transected from the proximal side [Figure 4C]. Traction of lymphatic tissue to the caudal side by assistant arm and traction of subclavian artery to the cranial side by assistant enabled sufficient lymphatic dissection close to the lower pole of the thyroid gland
Figure 4. Mediastinal dissection procedure under robot-assisted thoracoscopic esophagectomy in: the right upper mediastinum (A-E); the left upper mediastinum (F-J); the sub-aortic arch area (K); the sub-carinal area (L-N); the left pulmonary hilum area (O,P); and the lower mediastinum (Q,R). The robotic image is vertically and horizontally inverted by camera rotation to create a similar operative view to open thoracotomy. The left and upper sides of the monitor image are cranial and ventral, respectively. Encircled numeral corresponds to the port or arm number. The lymphatic tissue for dissection is encircled by a white dotted line. A: Aorta; AA: assistant arm; AV: azygos vein; D: diaphragm; E: esophagus; LH: left hand; LMB: left main bronchus; LRLN: left recurrent laryngeal nerve; MLN: metastatic lymph node; P: pericardium; PA: pulmonary artery; PB: pulmonary branch from vagal nerve; RH: right hand; RMB: right main bronchus; RRLN: right recurrent laryngeal nerve; S: suction; SCA: subclavian artery; T: trachea; TR: trachea retractor; VN: vagal nerve; TG: thyroid gland; LPV: left pulmonary vein; TD: thoracic duct.
Dissection of left RLN nodes
To dissect the left RLN lymph nodes, the esophagus was isolated by cutting the esophago-tracheal ligament. The esophagus was taped and extracted dorsally by assistant Cadiere held by Arm 2. The trachea was rotated with a tracheal retractor by the assistant. Lymph nodes situated on the left side of the trachea were extracted by cutting the ventral border of the lymph nodes. We used a vessel sealer to prevent accidental bleeding [Figure 4F]. After the identification of left RLN ventral to the esophagus, we transected the vessels’ flow into the left RLN lymph nodes at the level of aortic arch. We preserved several sympathetic cervical cardiac branches ventral to the left RLN. Then, we dissected the lymph nodes by exposing the left RLN with transecting the esophageal branches of left RLN from caudal to cranial side [Figure 4G]. Esophageal branches from the left inferior thyroid artery flow into the lymphatic tissue at the cervical-thoracic border. We transected these branches at their ventral inflow point by vessel sealer [Figure 4H]. After the isolation of left RLN, lymph nodes were retrieved by exposing the esophageal wall [Figure 4I and J]. The left main bronchus was extracted by trachea retractor to enlarge the subaortic area for dissection of the lymph nodes around the subaortic area to expose the dorsal side of left pulmonary artery [Figure 4K].
Dissection of subcarinal area
We exposed the right vagal nerve from cranial to caudal direction until the bronchial branches were branched from vagal nerve. The right bronchial artery was transected at its origin, and the peripheral branches of bronchial artery were transected at the cross point to vagal nerve. The vagal nerve was divided just distal of the branching point of pulmonary branches [Figure 4L]. Subcarinal lymph nodes were dissected to expose the pericardium and bilateral main bronchus [Figure 4M and N].
Dissection around left pulmonary hilum and lower mediastinum
To expose the left pulmonary hilum, we inserted a vessel sealer from cranial side and used it with the left hand [Figure 4O and P]. The usage of vessel sealer from cranial side enabled smooth dissection of left side of the mediastinum at middle and lower mediastinum [Figure 4Q]. For exhaustive dissection, we transected the thoracic duct and exposed the aortic wall by using a vessel sealer [Figure 4R].
Postoperative outcomes
We performed RATE in LDP in 58 cases from July 2018 to May 2021. The average robot set-up time including port placement, console time, and thoracic blood loss was 23.3 ± 7.6 min, 255 ± 64 min, and
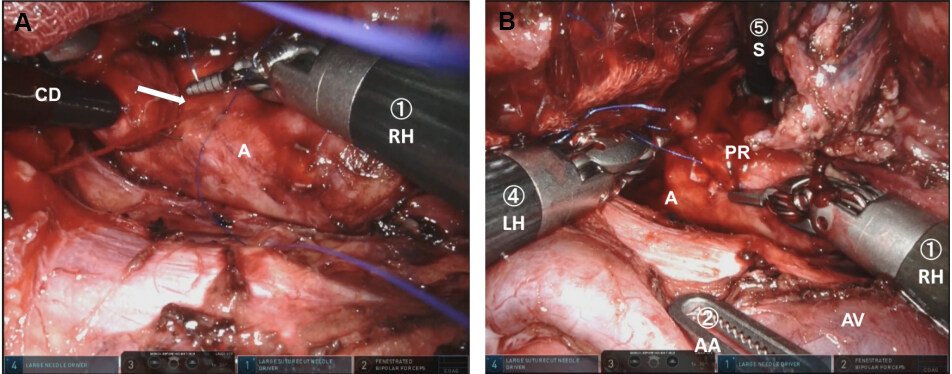
Figure 5. Hemostasis of bleeding from aorta by robotic platform: (A) direct suturing; and (B) hemostasis by ligation. Encircled numeral corresponds to the port or arm number. The arrow indicates the bleeding point. A: Aorta; AA: assistant arm; CD: cherry dissector; LH: left hand; PR: predjet; RH: right hand; S: suction; AV: azygos vein.
Surgical outcomes of the 58 patients who underwent robot-assisted thoracoscopic esophagectomy
| Operation time (min), mean ± SD | |
| Total | 609 ± 75 |
| Thorax | 309 ± 71 |
| Robot setup time | 23.3 ± 7.6 |
| Console time | 255 ± 64 |
| Amount of blood loss (mL), mean ± SD | |
| Total | 286 ± 187 |
| Thorax | 135 ± 126 |
| No. of dissected lymph nodes, mean ± SD | |
| Total | 58.3 ± 20.2 |
| Thorax | 27.1 ± 9.9 |
| Conversion to thoracotomy, No. (%) | 0 (0) |
| Curative resection according to pStagea, No. (%) | |
| pStage I (n = 18)b | 17 (94.4) |
| pStage II (n = 9) | 9 (100) |
| pStage III (n = 17) | 9 (100) |
| pStage IV (n = 14) | 8 (71.4) |
| Total (n = 58) | 51 (87.9) |
Postoperative complications with Clavien-Dindo classification grade over 2 included 11 (19.0%) pneumonia, 4 (6.9%) recurrent laryngeal nerve palsy, 2 (3.4%) chylothorax, 14 (24.1%) anastomotic leakage, and 4 (6.9%) acute respiratory distress syndrome. There were no specific complications related to the use of a robot. We experienced one surgical mortality by postoperative aortic bleeding after a non-curative operation for advanced esophageal cancer with extensive aortic invasion.
DISCUSSION
Camera rotation and manual hand control assignment in RATE in the LDP reproduced the surgical view and manipulation achieved in open esophagectomy and TE in the LDP. In this manuscript, we present the actual dissection procedure for esophageal cancer. We performed RATE in LDP for safe management of intraoperative emergency and meticulous dissection of the upper mediastinum because LDP is recognized as the optimal position for emergency thoracotomy in patients with serious tracheobronchial or major vessel injuries[27,28]. Wide dissection is necessary in the long mediastinum for the curative surgery in esophageal cancer, unlike pelvic surgery for rectal and gynecological malignancies. In esophageal cancer surgery, upper mediastinal dissection is considered as the most important procedure because of the high incidence of metastasis around bilateral RLN, which should be manipulated gently to prevent postoperative hoarseness and mis-swallowing[29]. Thus, we focused on the upper mediastinal dissection in TE or RATE. Insertion of dissection scissors by the surgeon’s right hand from the dorsal and caudal directions by TE in LDP enabled thorough dissection, especially in the left upper mediastinal region[27]. We can reproduce the dissection procedure of TE in LDP to perform RATE in LDP. Furthermore, the robotic platform has the advantage of enabling the surgeon to perform precise procedures in a deep narrow area such as the left side of the upper mediastinum due to the magnified three-dimensional view. Conversely, lower mediastinal dissection is easier with the patient in PP rather than in LDP because insertion of the dissection tool by the surgeon’s right hand can be possible from the cranial direction in PP. However, there are no sensitive nerves to be preserved in lower mediastinum, and the metastasis in lower mediastinum is infrequent compared to in upper mediastinum[29]. Therefore, the dissection of lower mediastinum can be easily performed by using the surgeon’s left hand in our procedure by RATE in LDP.
The most serious situation necessitating emergent thoracotomy include aorta and trachea-bronchial injury. We experienced one case of aortic bleeding by esophageal branch pull out during dissection around the aorta. This case was successfully managed without thoracotomy by direct suturing using 5-0 Prolene (Ethicon, Somerville, NJ, USA) with Cardiovascular Pledgets (Medtronic, Minneapolis, MN, USA). Only one additional port was inserted for the temporal hemostasis by compression with a cherry dissector. The robotic platform, with a three-dimensional view, articulation of instruments with seven degrees of freedom, tremor filtering, and hand control with a 3:1 motion scale, enabled direct suturing of the aorta and ligation with fine thread. Furthermore, the insertion of surgeon’s right hand from caudal direction was ergonomically ideal for direct suturing of the aorta. It is noteworthy that the surgeon can choose any EndWrist usage by manual hand control assignment in the case of repairing aorta and/or trachea-bronchial injury in robotic surgery.
Many surgeons prefer PP to LDP when performing thoracoscopic esophagectomy and RATE because an adequate surgical view can be easily obtained without the assistant’s effort. The limitation of RATE in LDP is the compressing of the lung and rotation of the trachea by an assistant using trachea retractor. Artificial pneumothorax by AirSeal insufflation system (ConMed) is useful in obtaining a good view without compressing the lung and diminishing the assistant’s efforts. However, trachea retraction by the assistant is mandatory to retrieve left RLN lymph nodes situated left side of the trachea. Two-lung ventilation in PP using artificial pneumothorax may be ideal to expose lymph nodes around the trachea because trachea rotation can be easily performed with a soft single lumen tube, unlike the hard double-lumen tube used for single-lung ventilation[30]. We will try RATE with two-lung ventilation in the prone position set by bed rotation for current LDP with anterior tilting. Emergent thoracotomy may be easily performed by restoring body position with bed rotation. This modification may improve the easiness of left RLN dissection and contribute to a more meticulous dissection with reducing the assistant’s effort. In addition, one of the ports for the assistant may be reduced.
CONCLUSION
Our results indicate that RATE in LDP to reproduce the surgical view and manipulation consistent with open esophagectomy is useful for performing upper mediastinal dissection to secure emergent thoracotomy.
DECLARATIONS
Authors’ contributionsPerformed operation: Ninomiya I, Okamoto K
Written the manuscript: Ninomiya I
Availability of data and materialsNot applicable.
Financial support and sponsorshipNone.
Conflicts of interestBoth authors declared that there are no conflicts of interest.
Ethical approval and consent to participateAll patients provided written informed consent before treatment, and the study was approved by the Ethics Committee of Kanazawa University Hospital. This work conforms to the guidelines set forth in the Helsinki Declaration of 1975 and later versions. This study was approved by Kanazawa University Hospital (Approval No: 2019-295, June 19, 2020).
Consent for publicationNot applicable.
Copyright© The Author(s) 2022.
REFERENCES
1. Biere SS, van Berge Henegouwen MI, Maas KW, et al. Minimally invasive versus open oesophagectomy for patients with oesophageal cancer: a multicentre, open-label, randomised controlled trial. Lancet 2012;379:1887-92.
2. Maas KW, Cuesta MA, van Berge Henegouwen MI, et al. Quality of life and late complications after minimally invasive compared to open esophagectomy: results of a randomized trial. World J Surg 2015;39:1986-93.
3. Yoshida N, Yamamoto H, Baba H, et al. Can minimally invasive esophagectomy replace open esophagectomy for esophageal cancer? Ann Surg 2020;272:118-24.
4. van Hillegersberg R, Boone J, Draaisma WA, Broeders IA, Giezeman MJ, Borel Rinkes IH. First experience with robot-assisted thoracoscopic esophagolymphadenectomy for esophageal cancer. Surg Endosc 2006;20:1435-9.
5. Weksler B, Sharma P, Moudgill N, Chojnacki KA, Rosato EL. Robot-assisted minimally invasive esophagectomy is equivalent to thoracoscopic minimally invasive esophagectomy. Dis Esophagus 2012;25:403-9.
6. van der Sluis PC, Ruurda JP, van der Horst S, et al. Robot-assisted minimally invasive thoraco-laparoscopic esophagectomy versus open transthoracic esophagectomy for resectable esophageal cancer, a randomized controlled trial (ROBOT trial). Trials 2012;13:230.
7. Huang L, Onaitis M. Minimally invasive and robotic Ivor Lewis esophagectomy. J Thorac Dis 2014;6 Suppl 3:S314-21.
8. Amaral M, Pimiento J, Fontaine JP. Robotic esophagectomy: the Moffitt Cancer Center experience. Ann Cardiothorac Surg 2017;6:186-9.
9. Egberts JH, Stein H, Aselmann H, Hendricks A, Becker T. Fully robotic da Vinci Ivor-Lewis esophagectomy in four-arm technique-problems and solutions. Dis Esophagus 2017;30:1-9.
10. Jin R, Xiang J, Han D, Zhang Y, Li H. Robot-assisted Ivor-Lewis esophagectomy with intrathoracic robot-sewn anastomosis. J Thorac Dis 2017;9:E990-3.
11. Nora I, Shridhar R, Meredith K. Robotic-assisted Ivor Lewis esophagectomy: technique and early outcomes. Robot Surg 2017;4:93-100.
12. van der Horst S, Weijs TJ, Ruurda JP, et al. Robot-assisted minimally invasive thoraco-laparoscopic esophagectomy for esophageal cancer in the upper mediastinum. J Thorac Dis 2017;9:S834-42.
13. Grimminger PP, Hadzijusufovic E, Ruurda JP, Lang H, van Hillegersberg R. The da Vinci Xi robotic four-arm approach for robotic-assisted minimally invasive esophagectomy. Thorac Cardiovasc Surg 2018;66:407-9.
14. He H, Wu Q, Wang Z, et al. Short-term outcomes of robot-assisted minimally invasive esophagectomy for esophageal cancer: a propensity score matched analysis. J Cardiothorac Surg 2018;13:52.
15. Zhang Y, Xiang J, Han Y, et al. Initial experience of robot-assisted Ivor-Lewis esophagectomy: 61 consecutive cases from a single Chinese institution. Dis Esophagus 2018:31.
16. Motoyama S, Sato Y, Wakita A, et al. Extensive lymph node dissection around the left laryngeal nerve achieved with robot-assisted thoracoscopic esophagectomy. Anticancer Res 2019;39:1337-42.
17. Na KJ, Park S, Park IK, Kim YT, Kang CH. Outcomes after total robotic esophagectomy for esophageal cancer: a propensity-matched comparison with hybrid robotic esophagectomy. J Thorac Dis 2019;11:5310-20.
18. Kim DJ, Hyung WJ, Lee CY, et al. Thoracoscopic esophagectomy for esophageal cancer: feasibility and safety of robotic assistance in the prone position. J Thorac Cardiovasc Surg 2010;139:53-59.e1.
19. Suda K, Ishida Y, Kawamura Y, et al. Robot-assisted thoracoscopic lymphadenectomy along the left recurrent laryngeal nerve for esophageal squamous cell carcinoma in the prone position: technical report and short-term outcomes. World J Surg 2012;36:1608-16.
20. Puntambekar S, Kenawadekar R, Kumar S, et al. Robotic transthoracic esophagectomy. BMC Surg 2015;15:47.
21. Somashekhar SP, Jaka RC. Total (transthoracic and transabdominal) robotic radical three-stage esophagectomy-initial indian experience. Indian J Surg 2017;79:412-7.
22. Osaka Y, Tachibana S, Ota Y, et al. Usefulness of robot-assisted thoracoscopic esophagectomy. Gen Thorac Cardiovasc Surg 2018;66:225-31.
23. Daiko H, Oguma J, Fujiwara H, et al. Robotic esophagectomy with total mediastinal lymphadenectomy using four robotic arms alone in esophageal and esophagogastric cancer (RETML-4): a prospective feasibility study. Esophagus 2021;18:203-10.
24. Shirakawa Y, Noma K, Maeda N, Tanabe S, Sakurama K, Fujiwara T. Microanatomy-based standardization of left upper mediastinal lymph node dissection in thoracoscopic esophagectomy in the prone position. Surg Endosc 2021;35:349-57.
25. Ninomiya I, Osugi H, Fujimura T, et al. Results of video-assisted thoracoscopic surgery for esophageal cancer during the induction period. Gen Thorac Cardiovasc Surg 2008;56:119-25.
26. Ninomiya I, Okamoto K, Yamaguchi T, et al. Optimization of robot-assisted thoracoscopic esophagectomy in the lateral decubitus position. Esophagus 2021;18:482-8.
27. Ninomiya I, Okamoto K, Fujimura T, Fushida S, Osugi H, Ohta T. Oncologic outcomes of thoracoscopic esophagectomy with extended lymph node dissection: 10-year experience from a single center. World J Surg 2014;38:120-30.
28. Markar SR, Wiggins T, Antonowicz S, Zacharakis E, Hanna GB. Minimally invasive esophagectomy: lateral decubitus vs. prone positioning; systematic review and pooled analysis. Surg Oncol 2015;24:212-9.
29. Udagawa H, Ueno M, Shinohara H, et al. The importance of grouping of lymph node stations and rationale of three-field lymphoadenectomy for thoracic esophageal cancer. J Surg Oncol 2012;106:742-7.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Ninomiya I, Okamoto K. Novel surgical technique of robot-assisted thoracoscopic esophagectomy in the lateral decubitus position. Mini-invasive Surg 2022;6:33. http://dx.doi.org/10.20517/2574-1225.2022.12
AMA Style
Ninomiya I, Okamoto K. Novel surgical technique of robot-assisted thoracoscopic esophagectomy in the lateral decubitus position. Mini-invasive Surgery. 2022; 6(6): 33. http://dx.doi.org/10.20517/2574-1225.2022.12
Chicago/Turabian Style
Ninomiya, Itasu, Koichi Okamoto. 2022. "Novel surgical technique of robot-assisted thoracoscopic esophagectomy in the lateral decubitus position" Mini-invasive Surgery. 6, no.6: 33. http://dx.doi.org/10.20517/2574-1225.2022.12
ACS Style
Ninomiya, I.; Okamoto K. Novel surgical technique of robot-assisted thoracoscopic esophagectomy in the lateral decubitus position. Mini-invasive. Surg. 2022, 6, 33. http://dx.doi.org/10.20517/2574-1225.2022.12
About This Article
Special Issue
Copyright
Data & Comments
Data

 Cite This Article 7 clicks
Cite This Article 7 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.