Ureter identification utilizing indocyanine green (ICG) imaging in colorectal surgery: a systematic review of the literature
Abstract
Aim: To describe the currently available evidence regarding the efficacy and safety of intraureteral indocyanine green (ICG) instillation for ureteral identification in colorectal surgery.
Methods: Systematic search of PubMed and Scopus through May 2022 reported according to PRISMA 2020. Studies reporting treatment of patients < 18 years, with unavailable full-text, reviews, editorials, animal studies, and studies including non-colorectal operations were excluded.
Results: Seven retrospective studies, published between 2020 and 2022, were identified, in which 142 patients (43% females) were evaluated, of which three were undertaken in the USA, two in Japan, and two in India. The most common indications for surgery were cancer in 58 patients (41%) and diverticular disease in 52 patients (36.6%). Most patients involved underwent robotic surgery (70%), while the remaining patients had laparoscopic surgery. The intraoperative ureteral injury was reported in one patient, while adverse effects (mainly transient hematuria) were reported in 10% of the study population (14 patients). The use of intrauretal ICG prolonged surgery by a median of 12.8 minutes.
Conclusion: Intraoperative visualization of ureters using ICG in colorectal surgery is safe and effective, according to the results of this study. However, this technique still bears the potential disadvantages of ureteral catheterization. Research is focusing on future dyes combining the ICG properties with renal excretion to minimize the need for stents. Further comparative studies are needed to reach safe results.
Keywords
INTRODUCTION
Ureteral injury is a potentially serious complication of colorectal surgery, with an incidence reaching up to 5%[1]. A understanding of the anatomy and careful dissection are of paramount importance to avoid such complications.
Ureteral injuries can be devastating. Moreover, 50%-70% of these injuries remain undiagnosed during the index surgery and are found later in the postoperative course, complicating their treatment and subjecting the patient to additional invasive procedures[1]. Obesity, extensive inflammation or locally advanced malignancies further increase the possibility of ureteral injury.
To help intraoperatively identify ureters, preoperative ureteral stent placement has been employed. Although stents can be palpated during open procedures, minimally invasive techniques with limited haptic feedback are used, but this technique is less reliable. Intraureteral indocyanine green (ICG) instillation to identify the ureters is another option, especially in the minimally invasive era. This review aims to discuss the currently available evidence regarding the efficacy and safety of intraureteral ICG instillation for ureters identification during colorectal surgery.
MATERIAL AND METHODS
Review registration
This study was reported in line with PRISMA 2020 guidelines[2]. Accordingly, this study was deemed exempt by our Institutional Review Board.
Search strategy and databases searched
A systematic search of PubMed and Scopus databases was undertaken through May 2022 by the first author (Z.G). The terms “ICG”, “indocyanine green”, “indocyanine”, “fluorescence”, “colorectal”, “colon”, “rectal”, “pelvic”, “ureter”, “ureteral”, “ureteric” and “urinary” combined with Boolean operators AND/OR to detect all available published studies related to the use of ICG for intraoperative ureter identification during colorectal surgery.
After eliminating duplicate studies, two authors (Z.G.) and (S.D.W.) independently screened the abstract list generated by the above search for potentially relevant studies. A full-text evaluation of the selected studies was undertaken for completeness and eligibility of reported data, based on our exclusion criteria.
Selection criteria
Studies deemed eligible for inclusion had to fulfill the following PICO criteria:
P (patients): adult patients undergoing colorectal surgery.
I (intervention): intraoperative ureter identification using indocyanine green (ICG) imaging.
C (comparator): none.
O (outcome): efficacy (number of intraoperative ureteral injuries) and procedure-related complications.
We excluded studies reporting treatment of patients younger than 18 years, studies with unavailable full-text, reviews, editorials, and animal studies.
Assessment of study quality and bias risk
The ROBINS-1 tool[3] was used to assess the risk of bias for all studies by two independent authors (Z.G. & S.D.W).
Outcomes
The primary outcome of this review was the efficacy of the ICG-guided intra-operative identification of ureters. Secondary outcomes included complications and adverse effects of this technology, along with operative times. The efficacy was assessed by the proportion of patients who suffered intraoperative ureteral injury despite the use of ICG ureteral imaging.
Data collection and analysis
Data of interest included the year and quality of the publication, number of patients, sex, efficacy of ICG-guided visualization, indication for surgery, type of surgery, previous operations, complications, duration surgery, and length of stay, among others. After a full-text evaluation of included studies, data were entered into Excel spreadsheets (Microsoft, Redmond, Washington, USA) and cross-checked by two authors (Z.G., S.D.W) for discrepancies.
Microsoft Excel was used to tabulate data. Cumulative analysis was performed, where possible. Categorical variables were extracted as numbers and reported as proportions.
RESULTS
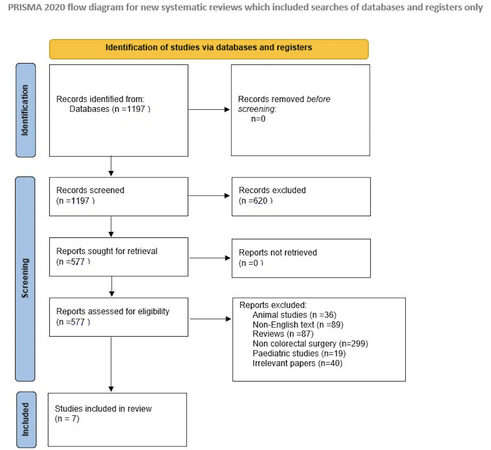
Seven studies[4-10], published between 2020 and 2022, that included 142 patients were identified. Figure 1 shows the study selection using the PRISMA flowchart. Three of the included studies were undertaken in the USA, two in Japan, and two in India [Table 1]. All studies were retrospective. Females represented almost 43% of the study population. The most common surgical indications were cancer in 58 patients (41%) and diverticular disease in 52 patients (36.6%). Most patients underwent robotic surgery (70%), while 30% had laparoscopic surgery. Only one patient suffered intraoperative ureteral injury despite the use of the ICG adjunct, while adverse effects were reported in 10% of the study population (14 patients). Etiology and type of surgery are demonstrated in Table 2. Reported adverse effects included mainly transient hematuria, as expected after ureteral catheterization [Table 3]. Four of the included studies reported data on the time needed for ureteral catheterization and ICG instillation, and the median duration was 12.8 minutes.
Characteristics of the included studies and patients’ demographics
| Study | Country | Type of study | Number of patients | Females(n) | ICG details | Ureteral stents |
| White et al., 2020[5] | USA | Prospective cohort study | 15 | 10 | 5 mL of 2.5 mg/mL ICG in each ureter | Yes (Bilateral) |
| Ryu et al., 2020[8] | Japan | Retrospective cohort study | 11 | 7 | N/A | Yes (Bilateral) |
| Kanabur et al., 2020[4] | USA | Retrospective case series | 5 | 0 | 5 mL of 2.5 mg/mL ICG in each ureter | Yes (Bilateral) |
| Mandovra et al., 2020[7] | India | Retrospective cohort study | 20 | N/A | 5-mg diluted in 2 mL of distilled water, flushed by 10ml of water in each ureter | Bilateral ureteric cannulation with catheter tip (Not indwelling) |
| Soriano et al., 2022[10] | USA | Retrospective cohort study | 83 | 43 | 5 mL of ICG followed by 5 mL of saline in each ureter | Bilateral ureteric cannulation with catheter tip (Not indwelling) |
| Hamada et al., 2022[6] | Japan | Retrospective case series | 6 | N/A | N/A | Yes (Bilateral) |
| Satish et al., 2022[9] | India | Retrospective case series | 2 | 1 | 2.5 mg (1 mL) of ICG dye diluted with 4 mL distilled water | Yes (Bilateral) |
Indication and type of surgery
| Study | Etiology | Preoperative radiation | Type of surgery |
| White et al., 2020[5] | Diverticulitis (7) Endometriosis (4) Cancer (3) IBD (1) | 2 | Robotic-assisted sigmoidectomy (9) Robotic-assisted low anterior resection (4) Robotic-assisted revision of J-pouch (2) Robotic-assisted left hemicolectomy (1) |
| Ryu et al., 2020[8] | Rectal cancer (11) Anal cancer (1) Rectal cancer recurrence (2) | 5 | Laparoscopic low anterior resection (4) Hartmann’s reversal (2) APR (5) Pelvic exenteration (1) |
| Kanabur et al., 2020[4] | Colorectal cancer (3) Colovesical fistula (1) Diverticular disease (1) | N/A | N/A |
| Mandovra et al., 2020[7] | N/A | N/A | Laparoscopic ventral mesh rectopexy (8) Laparoscopic posterior rectopexy (1) Laparoscopic LAR (6) Laparoscopic sigmoid colectomy (2) Laparoscopic right colectomy (2) Laparoscopic total colectomy (1) |
| Soriano et al., 2022[10] | Diverticulitis (43) Cancer (30) IBD (9) Chronic constipation (1) | 14 | Robotic left colectomy (1) Sigmoidectomy (46) LAR (24) APR (12) |
| Hamada et al., 2022[6] | Cancer (6) | N/A | Laparoscopic LAR + seminal vesicle resection (1) Tumor extirpation + Hartmann’s reversal (1) Posterior PE (1) APE with coccygectomy (1) Tumor extirpation with nephroureterectomy (1) Pelvic exenteration (1) |
| Satish et al., 2022[9] | Cancer (2) | 1 | Laparoscopic LAR and laparoscopic Hartmann’s |
Injuries and complications
White et al. reported in 16 patients who underwent complex robotic-assisted colorectal surgery that the instillation of ICG into the ureters with cystoscopy helped in safe and efficient ureter visualization[5]. ICG was administered through ureteral catheters. Intraureteral ICG was subsequently detected using near-infra-red laser fluorescence technology (Firefly®).
This preliminary report was followed by similar reports by Ryu et al., Kanabur et al., and
In 2022, Soriano et al. published a modification of this technique: ICG injection into the ureters for fluorescent without placing a ureteral catheter[10]. This procedure without the placement of indwelling catheters was four times faster than the conventional method. Additionally, transient hematuria was seen in 50% (10/20 patients) of cases that had stent placement versus 3% (2/63) in the simple injection group
Finally, Satish et al., along with Hamada et al., reported also in 2022 another six cases in total of successful intraoperative identification of the ureters using ICG technology in complicated colorectal surgery without any adverse effectssup[6,9].
Assessment of risk of bias
All of the studies were assessed as high risk for bias according to the ROBINS-I tool [Table 4].
ROBINS-I assessment of bias
| Included studies | Bias due to confounding | Bias in selection of participants in the study | Bias in classification of intervention | Bias due to deviation from the intended intervention | Bias due to missing data | Bias in measurement of outcomes | Bias in selection of the reported result | Overall risk |
| White et al., 2020[5] | Moderate | High | Low | Low | Low | Low | Moderate | High |
| Ryu et al., 2020[8] | Moderate | High | Low | Low | Low | Low | Moderate | High |
| Kanabur et al., 2020[4] | Moderate | High | Low | Low | Low | Low | Moderate | High |
| Mandovra et al., 2020[7] | Moderate | High | Low | Low | Low | Low | Moderate | High |
| Soriano et al., 2022[10] | Moderate | High | Low | Low | Low | Low | Moderate | High |
| Hamada et al., 2022[6] | Moderate | High | Low | Low | Low | Low | Moderate | High |
| Satish et al., 2022[9] | Moderate | High | Low | Low | Low | Low | Moderate | High |
DISCUSSION
Abdominal and pelvic operations can be complicated by ureteral injury in up to 10% of cases[1]. Ureteral injury can be caused by either ligation, division, devascularization, thermal injury or laceration[11]. Ureteral injuries can be quite devastating, raising treatment costs and morbidity and with long sequelae of adverse events while trying to repair the initial damage. Unfortunately, 50%-70% of these injuries remain undiagnosed during the index surgery and are found later in the postoperative course, complicating treatment and subjecting the patient to additional invasive procedures[1]. Obesity, extensive inflammation or locally advanced malignancies further increase the possibility of ureteral injury. Several methods have been developed over time for intraoperative ureteral identification.
Conventional ureteral stents
Traditionally, one of the tools in the surgical armamentarium for identifying the ureter intraoperatively has been the preoperative or intraoperative insertion of endoluminal stents that allows facilitates identification with the assistance of haptic feedback. Cystoscopic insertion of ureteral stents is a standard procedure used as an adjunct to intraoperatively identify the ureters and to aid in the avoidance of injuries[12]. Furthermore, prophylactic stent insertion helps in the prompt intraoperative identification of a ureteral injury, rather than delayed postoperative recognition. Conversely, ureteral catheters can be associated with longer operative times, increased costs and possible adverse events such as ureteral injury, acute kidney injury, and infectious complications[13]. A recent meta-analysis by Hird et al., including 98,507 patients from nine retrospective cohort studies, did not demonstrate a significant difference in the number of events of ureteral injury among patients who underwent prophylactic stent insertion compared to those individuals in whom stents were not inserted[14]. However, the studies included in this meta-analysis were retrospective and non-randomized. Thus, there is a high risk of confounding bias due to patient selection for stent insertion according to the indication and complexity of each case. In other words, patients at higher risk for ureteral injury may have undergone stent placement more often than patients in whom surgery is deemed less uncomplicated. To date, no randomized-control studies have been published on the prophylactic insertion of ureteral stents in colorectal surgery.
Lighted ureteral stents
With the evolution of minimally invasive surgery, the tactile feedback provided by the stents was removed. Therefore, lighted stents started to be explored to overcome this issue. In a recent systematic review by Mazzarella et al., 3/743 (0.4%) patients had intraoperative ureteral injuries, and the authors concluded that placement of lighted ureteral stents (LUS) helps to intraoperatively safeguard and identify the ureteral injury[15]. In the same study, the authors also reported hematuria in 97.6% of patients, but it was spontaneously resolved. The authors noted that the transient hematuria was due to the preoperative placement of LUS. Despite the initial enthusiasm, lighted stents are still an interventional procedure bearing the same risks as conventional stents, such as longer operative times, increased costs and possible adverse events such as ureteral injury, acute kidney injury, and infectious complications. Furthermore, they may potentially introduce additional risks, with reports of external burns to patients and surgical drape burns[4]. Furthermore, they are not easily available.
Methylene Blue
Methylene blue is a dye that has been used in humans during surgery for localization procedures with a good proven safety profile. This dye has near-infrared fluorescent properties that were discovered nearly 10 years ago[16]. Methylene blue, safe to administer intravenously, is excreted through the kidneys and is concentrated in urine[17,18]. Methylene blue can be excited at 668 nm and emits light in the near-infrared region[18]. Since methylene blue is mainly excreted through the kidneys, urine becomes near-infrared fluorescent after a single intravenous dose, permitting real-time ureteral visualization[19]. The main advantage of this technique is that no ureteral catheterization is necessary, minimizing potential infectious and traumatic complications. However, this dye, although safe to use in low doses (< 2 mg/kg), can cause severe adverse effects, including anaphylaxis, methemoglobinemia, or hemolysis, in patients with G6PD deficiency[19]. In addition, intravenous administration of methylene blue creates an artificial decrease in oxygen saturation when measured by pulse oximetry, and this effect can be continued for several minutes. Methylene blue is excreted by the kidneys; therefore, its use is limited by the status of renal function in each patient. An additional limitation is that it is restricted to patients who can convert methylene blue into non-fluorescing leucomethylene blue, caused by the reduction and/or acidity of the environment[18].
Indocyanine Green (ICG) Imaging
ICG fluorescence imaging was introduced into minimal invasive surgery (MIS) to provide surgeons with a better view and anatomical information intraoperatively[20-25]. In colorectal surgery, in particular, this has been used to provide information regarding bowel perfusion, aimed at reducing anastomotic leaks[26]. ICG is intravenously administered, with no adverse effects except for possible allergy to the dye. The dye is then present in bile (aiding in the biliary system visualization), vessels and lymph nodes. ICG is excreted by bile when intravenously administered intravenously, so for the ureters to be visualized, cystoscopy-guided ICG instillation must be performed. This dye has the unique property of binding to and staining the proteins of the ureteral epithelium for the entire procedure (although reversibly)[27,28].
Based on the results of our review, ICG is a safe way of intraoperative ureter visualization that overcomes the possible side effects of methylene blue administration. Nevertheless, this technique still requires cystoscopy and intraureteral dye instillation, raising the risk for infectious complications and ureteral injury. Furthermore, this increases operative times and costs.
Experimental dyes
● IRDye CW800
Three studies report results on CW800-CA[29-31] and one reports on CW800-BK[32]. These two dyes were intravenously injected into pigs. All doses were below the results from initial toxicology reports[26]. Ureters were visualized within 10 min after administration and remained visible for 120 min. In three other studies, ureters could be visualized at any dose[22,24,25]. IRDye CW800 is currently being studied in several ongoing phase II studies. Safety, efficacy and dosing of CW800-BK for ureter visualization and identification are being studied in two ongoing trials (NCT03387410, NCT03106038).
● ZW800-1
Another experimental dye, cRGD-ZW800-1, was tested in rats[33]. This dye is metabolized by the kidneys. Low dosages of this experimental dye were used intravenously in rats. One animal study reports ureter visualization in rats using cRGD-ZW800-1[33]. The ureters were visualized 10 minutes after injection and remained visible for 8 hours. No adverse effects have been reported in doses up to 5.0 mg/kg in rats [34]. Currently, there is one ongoing clinical study regarding the use of ZW800-1 for ureter identification (2017-001954-32).
● Liposomal ICG
This chemical form of ICG increases its clearance by the kidneys, enabling ureter visualization without intraureteral instillation. Two animal studies reported the use of liposomal ICG for ureter visualization[35,36]. Toxicity levels are not available for liposomal ICG per se, but for each component of this composite dye. No ongoing clinical trials have been identified.
● UreterGlow and sodium fluorescein
We previously reported two other new near-infrared fluorescent dyes: UreterGlow[37], which can be injected systemically but is excreted primarily through the renal system, and sodium fluorescein[38], which is already used in ophthalmology and intravenous angiography. As previously mentioned, both dyes are mainly excreted via the renal system. Ureters were visible with NIR fluorescence camera when using UreterGlow and at 530 nm wavelength mode when using sodium fluorescein. In rats, after sodium fluorescein intravenous infusion, fluorescence visualization of the ureters was noted 5-10 min following kidney visualization[38]. UreterGlow was injected intravenously into Yorkshire pigs, and peristalsis of the ureter could be immediately observed. The most peripheral part of the ureter could be visualized under NIR 60 min after initial injection and remained visible for > 2 h[37].
● Other new dyes
There are some reports on other dyes, but they include only scarce data and lack toxicity levels, and only in animal studies[39-41].
LIMITATIONS
Our study has several limitations. First, the studies included were not comparative. In addition, we have to acknowledge possible selection bias, as usually intraoperative ureter visualization may have been preferentially used in complicated cases. Although the studies assessed the time needed for stent placement and ICG instillation, they did not take into account the time potentially saved with the direct ureter visualization in complicated cases. Furthermore, all studies were deemed as high risk of bias according to the ROBINS-I tool, thus lowering the evidence provided by our study. According to our assessment, the results of our study may be biased and caution is needed in making judgments about the efficacy of intraoperative ureter visualization with the use of ICG. Finally, only one study compared patients with both stent placement and ICG instillation versus simple ICG instillation. All others reported results on patients with simultaneous ureteral stent placement and intraureteral ICG infusion, increasing the heterogeneity of the reported results.
CONCLUSION
Intraoperative visualization of ureters using ICG in MIS colorectal surgery is safe and effective, according to the results of this study. However, this technique still bears the potential disadvantages of ureteral catheterization. Research is focusing on future dyes combining the ICG properties with renal excretion to minimize the need for stents. Further comparative studies are needed prior to more widespread adoption.
DECLARATIONS
Authors’ contributionsMade substantial contributions to the conception and design of the study and performed data analysis and interpretation: Wexner SD, Garoufalia Z
Performed data acquisition, as well as providing administrative, technical, and material support Wexner SD, Garoufalia Z
Availability of data and materialsNot applicable.
Financial support and sponsorshipNone.
Conflicts of interestWexner SD receives royalties for intellectual property from Medtronic, Intuitive and Karl Storz Endoscopy and receives consulting fees for consulting from Medtronic, Intuitive, and Stryker.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Author(s) 2022.
REFERENCES
2. Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. PLoS Med 2021;18:e1003583.
3. Sterne JA, Hernán MA, Reeves BC, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ 2016;355:i4919.
4. Kanabur P, Chai C, Taylor J. Use of indocyanine green for intraoperative ureteral identification in nonurologic surgery. JAMA Surg 2020;155:520-1.
5. White LA, Joseph JP, Yang DY, et al. Intraureteral indocyanine green augments ureteral identification and avoidance during complex robotic-assisted colorectal surgery. Colorectal Dis 2021;23:718-23.
6. Hamada M, Matsumi Y, Sekimoto M, Kurokawa H, Kita M, Kinoshita H. Image navigation surgery with the fluorescent ureteral catheter of recurrent tumors in the pelvic cavity. Dis Colon Rectum 2022;65:e72-6.
7. Mandovra P, Kalikar V, Patankar RV. Real-time visualization of ureters using indocyanine green during laparoscopic surgeries: can we make surgery safer? Surg Innov 2019;26:464-8.
8. Ryu S, Hara K, Kitagawa T, et al. Fluorescence vessel and ureter navigation during laparoscopic lateral lymph node dissection. Langenbecks Arch Surg 2022;407:305-12.
9. Satish V N V R, Acharya A, Ramachandran S, Narasimhan M, Ardhanari R. Fluorescent ureterography with indocyanine green in laparoscopic colorectal surgery: a safe method to prevent intraoperative ureteric injury. J Minim Access Surg 2022;18:320-3.
10. Soriano CR, Cheng RR, Corman JM, Moonka R, Simianu VV, Kaplan JA. Feasibility of injected indocyanine green for ureteral identification during robotic left-sided colorectal resections. Am J Surg 2022;223:14-20.
11. Hughes ES, McDermott FT, Polglase AL, Johnson WR. Ureteric damage in surgery for cancer of the large bowel. Dis Colon Rectum 1984;27:293-5.
12. Coakley KM, Kasten KR, Sims SM, Prasad T, Heniford BT, Davis BR. Prophylactic ureteral catheters for colectomy: a national surgical quality improvement program-based analysis. Dis Colon Rectum 2018;61:84-8.
13. Hassinger TE, Mehaffey JH, Mullen MG, et al. Ureteral stents increase risk of postoperative acute kidney injury following colorectal surgery. Surg Endosc 2018;32:3342-8.
14. Hird AE, Nica A, Coburn NG, Kulkarni GS, Nam RK, Gien LT. Does prophylactic ureteric stenting at the time of colorectal surgery reduce the risk of ureteric injury? Colorectal Dis 2021;23:1060-70.
15. Mazzarella G, Muttillo EM, Picardi B, Rossi S, Rossi Del Monte S, Muttillo IA. Real-time intraoperative ureteral identification in minimally invasive colorectal surgery: a systematic review. J Laparoendosc Adv Surg Tech A 2022;32:627-33.
16. Matsui A, Tanaka E, Choi HS, Kianzad V, Gioux S, Lomnes SJ, Frangioni JV. Real-time, near-infrared, fluorescence-guided identification of the ureters using methylene blue. Surgery 2010;148:78-86.
17. Stolik S, Delgado J, Pérez A, Anasagasti L. Measurement of the penetration depths of red and near infrared light in human “ex vivo” tissues. J Photoch Photobiol B: Biology 2000;57:90-3.
18. Barnes TG, Hompes R, Birks J, et al. Methylene blue fluorescence of the ureter during colorectal surgery. Surg Endosc 2018;32:4036-43.
19. Peter C, Hongwan D, Küpfer A, Lauterburg BH. Pharmacokinetics and organ distribution of intravenous and oral methylene blue. Eur J Clin Pharmacol 2000;56:247-50.
20. Schols RM, Connell NJ, Stassen LP. Near-infrared fluorescence imaging for real-time intraoperative anatomical guidance in minimally invasive surgery: a systematic review of the literature. World J Surg 2015;39:1069-79.
21. Schaafsma BE, Mieog JS, Hutteman M, et al. The clinical use of indocyanine green as a near-infrared fluorescent contrast agent for image-guided oncologic surgery. J Surg Oncol 2011;104:323-32.
22. Schols RM, Bouvy ND, van Dam RM, Stassen LP. Advanced intraoperative imaging methods for laparoscopic anatomy navigation: an overview. Surg Endosc 2013;27:1851-9.
23. Boni L, David G, Mangano A, et al. Clinical applications of indocyanine green (ICG) enhanced fluorescence in laparoscopic surgery. Surg Endosc 2015;29:2046-55.
24. Baiocchi GL, Diana M, Boni L. Indocyanine green-based fluorescence imaging in visceral and hepatobiliary and pancreatic surgery: state of the art and future directions. World J Gastroenterol 2018;24:2921-30.
25. Verbeek FP, Schaafsma BE, Tummers QR, et al. Optimization of near-infrared fluorescence cholangiography for open and laparoscopic surgery. Surg Endosc 2014;28:1076-82.
26. Blanco-Colino R, Espin-Basany E. Intraoperative use of ICG fluorescence imaging to reduce the risk of anastomotic leakage in colorectal surgery: a systematic review and meta-analysis. Tech Coloproctol 2018;22:15-23.
27. Alander JT, Kaartinen I, Laakso A, et al. A review of indocyanine green fluorescent imaging in surgery. Int J Biomed Imaging 2012;2012:940585.
29. Schols RM, Lodewick TM, Bouvy ND, van Dam GM, Dejong CH, Stassen LP. Application of a new dye for near-infrared fluorescence laparoscopy of the ureters: demonstration in a pig model. Dis Colon Rectum 2014;57:407-11.
30. Korb ML, Huh WK, Boone JD, et al. Laparoscopic fluorescent visualization of the ureter with intravenous IRDye800CW. J Minim Invasive Gynecol 2015;22:799-806.
31. Al-Taher M, van den Bos J, Schols RM, Kubat B, Bouvy ND, Stassen LPS. evaluation of a novel dye for near-infrared fluorescence delineation of the ureters during laparoscopy. BJS Open 2018;2:254-61.
32. Marshall MV, Draney D, Sevick-Muraca EM, Olive DM. Single-dose intravenous toxicity study of IRDye 800CW in Sprague-Dawley rats. Mol Imaging Biol 2010;12:583-94.
33. Verbeek FP, van der Vorst JR, Tummers QR, et al. Near-infrared fluorescence imaging of both colorectal cancer and ureters using a low-dose integrin targeted probe. Ann Surg Oncol 2014;21 Suppl 4:S528-37.
34. Handgraaf HJM, Boonstra MC, Prevoo HAJM, et al. Real-time near-infrared fluorescence imaging using cRGD-ZW800-1 for intraoperative visualization of multiple cancer types. Oncotarget 2017;8:21054-66.
35. Portnoy E, Nizri E, Golenser J, Shmuel M, Magdassi S, Eyal S. Imaging the urinary pathways in mice by liposomal indocyanine green. Nanomedicine 2015;11:1057-64.
36. Friedman-Levi Y, Larush L, Diana M, et al. Optimization of liposomal indocyanine green for imaging of the urinary pathways and a proof of concept in a pig model. Surg Endosc 2018;32:963-70.
37. Dip FD, Nahmod M, Anzorena FS, et al. Novel technique for identification of ureters using sodium fluorescein. Surg Endosc 2014;28:2730-3.
38. Mahalingam SM, Dip F, Castillo M, et al. Intraoperative ureter visualization using a novel near-infrared fluorescent dye. Mol Pharm 2018;15:3442-7.
39. Meershoek P, KleinJan GH, van Oosterom MN, et al. Multispectral-fluorescence imaging as a tool to separate healthy from disease-related lymphatic anatomy during robot-assisted laparoscopy. J Nucl Med 2018;59:1757-60.
40. Rowe CK, Franco FB, Barbosa JA, et al. A novel method of evaluating ureteropelvic junction obstruction: dynamic near infrared fluorescence imaging compared to standard modalities to assess urinary obstruction in a swine model. J Urol 2012;188:1978-85.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Garoufalia Z, Wexner SD. Ureter identification utilizing indocyanine green (ICG) imaging in colorectal surgery: a systematic review of the literature. Mini-invasive Surg 2022;6:51. http://dx.doi.org/10.20517/2574-1225.2022.60
AMA Style
Garoufalia Z, Wexner SD. Ureter identification utilizing indocyanine green (ICG) imaging in colorectal surgery: a systematic review of the literature. Mini-invasive Surgery. 2022; 6: 51. http://dx.doi.org/10.20517/2574-1225.2022.60
Chicago/Turabian Style
Garoufalia, Zoe, Steven D. Wexner. 2022. "Ureter identification utilizing indocyanine green (ICG) imaging in colorectal surgery: a systematic review of the literature" Mini-invasive Surgery. 6: 51. http://dx.doi.org/10.20517/2574-1225.2022.60
ACS Style
Garoufalia, Z.; Wexner SD. Ureter identification utilizing indocyanine green (ICG) imaging in colorectal surgery: a systematic review of the literature. Mini-invasive. Surg. 2022, 6, 51. http://dx.doi.org/10.20517/2574-1225.2022.60
About This Article
Copyright
Data & Comments
Data
 Cite This Article 30 clicks
Cite This Article 30 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.