The effectiveness of the magnetic ring (LINX®) Device in managing reflux after sleeve gastrectomy; long term results: a seven-years' experience
Abstract
Aim: The magnetic ring (LINX®) system was approved by the FDA in 2012 as a device to augment the lower esophageal sphincter pressure to manage reflux in the general population. It was introduced into the bariatric population in 2014 as an alternative to Roux-en-Y gastric bypass (RYGB) in managing reflux after sleeve gastrectomy (SG).
Materials: Between January 2015 and December 2021, Seventeen patients had the Magnetic Ring (MR) device placement to manage their reflux after SG.
Methods: The mean age was 54 ± 12 years. Their mean body mass index (BMI) was 31 ± 6 kg/m2. The mean time between the SG and MR device placement was 41 ± 19 months. The mean DeMeester score was 48 ± 26
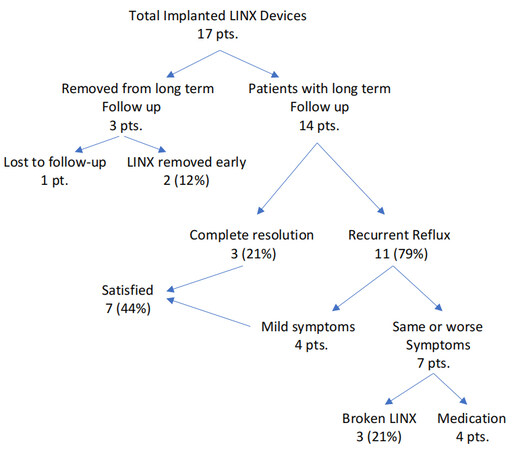
Results: All seventeen patients underwent a successful laparoscopic placement of the MR device. Two patients had early post-operative dysphagia requiring removal of the device and one patient was lost to follow-up. The mean follow up of the remaining fourteen patients was 65 ± 31 months. Recurrent reflux occurred in 11 (79%). In three of these eleven (27%) patients the recurrent reflux was due to a broken device. None of these patients were interested in conversion to RYGB.
Conclusion: The MR device is a valuable tool in treating reflux after SG in spite of its potential risk of recurrence. Our results can be interpreted as being dissatisfying if complete resolution is the goal. Larger studies, with more than 3 year follow up, are needed before a final decision is made on its value in the bariatric population.
Keywords
INTRODUCTION
The success of managing reflux using the MR device in 2012 in the general population after the FDA approval triggered the interest of a handful of bariatric surgeons to use this device in managing reflux after SG[1,2]. The system depends on the magnetic characteristics of its beads in augmenting the lower esophageal sphincter pressure to control the reflux [Figure 1]. We introduced this system into our practice in 2014 to manage reflux in the general population. As bariatric surgeons, we were treating reflux after SG using anterior or posterior fundoplication when the fundus of the stomach was dilated[3]. Our problem, however, was in managing this reflux after SG when the fundus of the stomach was not dilated enough to allow a fundoplication to be done while the patients were refusing the conversion to RYGB. We found in the MR device the solution to this problem.
METHODS
Our first patient to have the MR device (ETHICON J&J Cincinnati, OH) placement to manage her reflux after SG was done in January 2015[4]. Since then and until December 2021, we implanted the MR device in seventeen patients to manage their reflux after SG, including fourteen females and three males, with a mean age of 54 ± 12 years. Their initial mean weight at the time of placement of the MR device was 180 ± 45 lbs. with a mean BMI of 31 ± 6 kg/m2. The mean time between their SG and the placement of the MR device was 41 ± 19 months. Our endpoint was complete resolution of reflux after > 3 years of implantation.
All patients had pre-operative Barium swallow, high-resolution esophageal motility and upper endoscopy with 48-hour pH monitoring using the Bravo® capsule (GIVIN imaging, Duluth, GA). Their mean DeMeester score was 48 ± 26 (normal < 14.72). On a scale of 1-10 (10 being the worst reflux), the personal perceived severity of reflux score was 7 ± 3. Six (35%) patients had esophagitis. Twelve patients had a hiatal hernia < 2 cm and two patients had a 3 cm hiatal hernia. Sixteen (94%) patients had reflux before their SG, while only one (6%) patient had De Novo reflux 36 months after SG. All patients were given the Gastro Esophageal Reflux Disease-Health-Related Quality of Life survey (GERD-HRQL) to evaluate the severity of their symptoms. Their average pre-operative GERD-HRQL score was 46/75 ± 19/75.
The hospital Institutional Review Board (IRB) deemed this study exempt as these patients were in the registry of the local Michigan Bariatric Surgery Collaborative (MBSC) and the national The Metabolic and Bariatric Surgery Accreditation and Quality Improvement Program (MBSAQIP) of the American College of Surgeons.
Statistical analysis
Descriptive statistics will be calculated to characterize the study group. Continuous variables will be described using the mean with standard deviation or median with range, depending upon the distribution of the data. Categorical variables will be described as frequency distributions. Pre- to post-changes will be evaluated using the paired t-test or the McNemar chi-squared test. All data will be analyzed using SPSS v. 28.0 and a P-value of 0.05 or less will be considered to indicate statistical significance.
Operative description
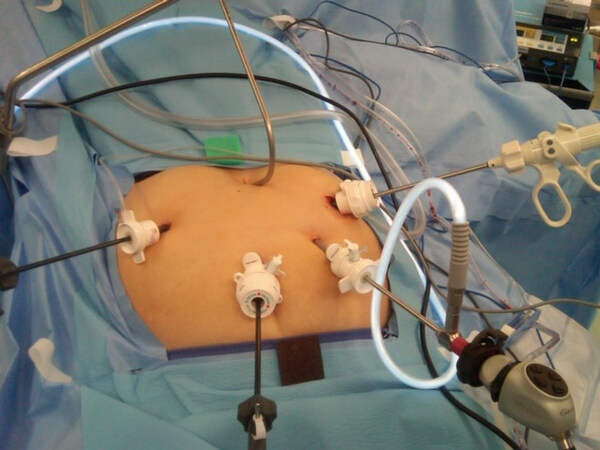
The procedure was performed using four trocars through five incision sites. These trocars were positioned in the same locations as the trocars positioning for the initial SG [Figure 2]. All patients underwent laparoscopic lysis of adhesions due to the previous SG. The esophagus was mobilized and brought into the abdominal cavity for a distance of 2-4 cm. The esophagus was then sized at the gastroesophageal junction without any tubes inside its lumen. The selected MR device size to be placed was two sizes above the size of the esophagus. In all patients, we approximated the posterior crus of the diaphragm allowing for an opening to pass a 48-Frbougie without difficulty. This corrected the presence of the HH and decreased the size of the hiatus. We also performed gastric plication to decrease the size of the dilated proximal stomach body and fundus when present. This dilation of the proximal stomach was not enough to be used as a fundoplication, either anterior or posterior. The gastric plication was also done to help the patients who were experiencing some weight regain, due to the dilation, as a re-sleeve to decrease their food intake.
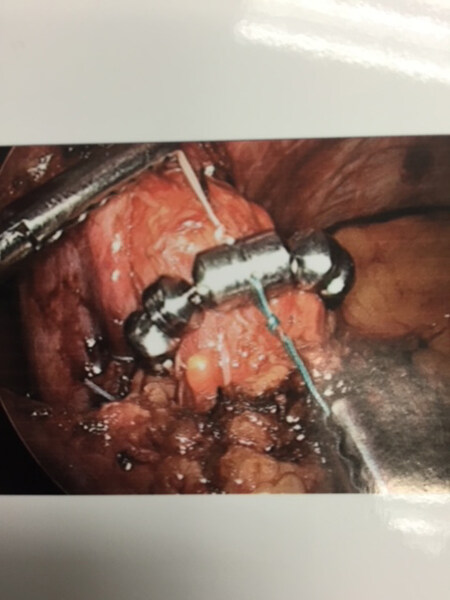
The bougie was then removed and the MR device was introduced through the left upper quadrant 10 mm trocar. The device was then placed around the esophagus through a window in front of the posterior vagus nerve. Care was taken not to injure the anterior or posterior vagus nerves. The buckle was then secured [Figure 3].
RESULTS
All seventeen patients underwent successful laparoscopic placement of the MR device without intra-operative complications or conversion to an open procedure. The average size of the implanted MR device was 15 ± 1 beads, with a range of 13-17 beads. Ten patients received gastric plication. The mean operative time was 83 ± 27 min.
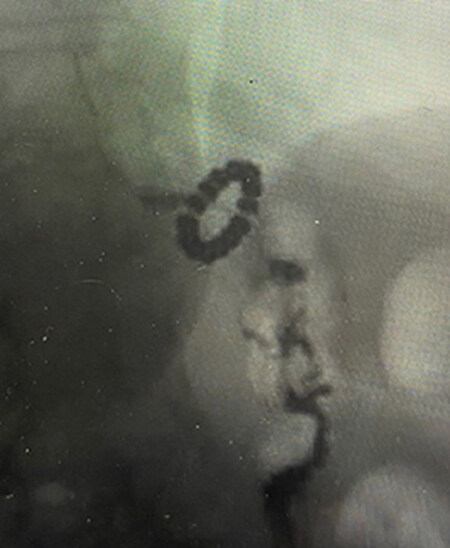
The patients were started on a regular diet on the same day of surgery. They were instructed to have a small meal every two hours to exercise the MR device beads in their position. A Gastrographin UGI contrast study was performed on the morning of post-operative day one [Figure 4]. The patients were then discharged home if they were tolerating their diet. The mean length of hospital stay was 1.2 ± 0.6 days.
Three patients were excluded from further analysis of their data and long-term follow-up. Two (12%) of these patients developed severe post-operative dysphagia requiring early MR device removal. One of these two patients had her device removed after 20 days. The second patient required esophageal dilation to stretch the device within three weeks of placement. She was found later to have erosion of the device requiring removal within three months. The third patient was lost to follow-up.
The remaining fourteen patients had long-term follow-ups between 3-7 years with a mean follow-up of
Of the 14 patients, 12 completed the five-year follow-up, and their five-year GERD-HRQL score went up to 19/75 ± 14/75, which was much higher compared to their one-year score (8/75 ± 7/75). However, when we compared their score before the placement of the MR device and at five years, we found the score to be still lower than the original pre-MR device placement. This comparison showed a > 50% drop in the original score, which was a statistically significant difference in spite of the patients’ recurrent symptoms
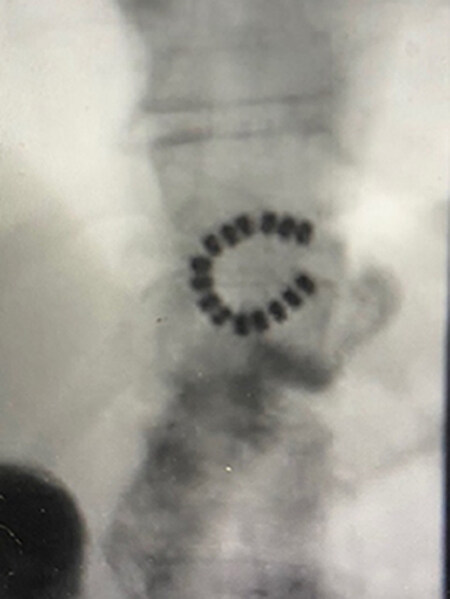
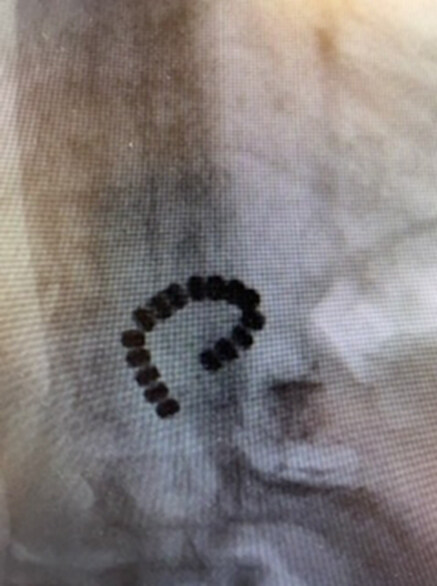
Three of the eleven (27%) patients had recurrent reflux due to a broken connection between the beads leading to an open MR device [Figures 5 and 6]. The broken devices were found accidentally on a chest X-ray or UGI study for follow-up. This complication occurred in 3/14 patients (21%). None of these patients had an MRI. The average time between the placement of the MR device and the finding that the device was broken was 4.7 ± 0.6 years. One patient had the broken MR device removed without conversion to RYGB to control her reflux. She elected to stay on medication. The other two patients elected to keep the broken MR device in place and stay on the medication that was controlling their symptoms.
Complete resolution of reflux occurred in 3/14 (21%) patients. All three patients were off PPI. There was no difference in the BMI of patients with recurrent reflux vs. those with successful control of their reflux
Patient satisfaction after one year of the fourteen patients was 100%. However, this satisfaction dropped after 3-7 years of follow-up to only 50% (7/14 patients). This included the three patients with complete resolution of symptoms and four patients with mild recurrence of symptoms which was easily controlled with less medication. If we include the two patients who had early device removal, the overall satisfaction will drop to 44% (7/16 patients). Overall, 13/16 (81%) patients had to go back on medication to manage their reflux [Figure 7]. None of the patients with recurrent reflux elected to have a conversion to RYGB and they were satisfied to stay on medication.
DISCUSSION
The standard recommendation for managing reflux after SG is conversion to RYGB[6]. Patients who originally chose SG over RYGB to avoid the metabolic complications of the RYGB are finding themselves being forced into choosing what they declined originally in order to manage their reflux after SG. In addition, reflux after RYGB is a well-known complication that can occur in 20%-33% of patients[7,8]. Actually, in a study on the use of anti-reflux medications after one year of RYGB, the authors found that around 44% of patients were still using these anti-reflux medications[9]. When comparing the initial RYGB to the conversion from SG to RYGB procedure, there is an increased risk of intraoperative bleeding and increased operative time in the conversion procedures. In addition, the conversion procedures required longer intensive care unit stays and longer hospital stays[10]. This indicates that the conversion to RYGB, with its high risk of surgical complications and metabolic derangement, may not be the best option after all.
The success of the magnetic ring (LINX®) system in managing reflux in the general population gave new hope to the subgroup of patients who were suffering from reflux after SG and did not want to undergo a conversion to RYGB to treat their reflux. The FDA, however, listed any previous esophageal or gastric surgery as a precaution since these patients were not studied by the company. The FDA left the decision of using this device in this group of patients to the judgment of the surgeon and the acceptance of the patient after being informed about its risks and complications. Being among the early adaptors of using this device in the general population and as a bariatric surgeon at the same time who was faced with the problem of managing reflux after SG, we found a solution by using the MR device. This was helpful, especially in patients who did not have much of a dilated proximal gastric pouch where fundoplication could be performed. We did offer this option to few patients after getting their consent.
The first publication describing using the MR device in managing reflux after SG was in 2015[2]. This was the year when we started to perform this procedure, as well, not knowing that other surgeons had started this already. Following this, we published our own case report of one-year follow-up, in addition to a case report on using the MR device to manage reflux after RYGB[4,11].
Our results may not be very satisfying (overall, 56% patient dissatisfaction), especially when 81% of patients had some type of recurrent reflux symptoms and going back on medication. We viewed any recurrence of reflux as a failure of the MR device. However, when looking at the GERD-HRQL scores at five years, we see a significant drop in the scores, indicating that some type of control of the reflux symptoms is enough for the patients to avoid the conversion to RYGB and be able to control their symptoms with less medication. If we use this reduction in the GERD-HRQL and the reduction of medication use as a successful outcome similar to other studies, then we can say that the use of the MR device was successful in controlling the reflux post SG[12].
The alarming problem is that disconnection of these devices is found after 4-5 years, which makes their long-term effectiveness questionable and may deter some patients from seeking this approach unless modifications are made to these devices. It is important to note that all these broken devices were implanted before the company issued a recall on them due to recognizing the possibility of this separation to occur [Figure 8]. In April 2018, the FDA issued a recall “Class 2 Device Recall LINX Reflux Management System” that was terminated on November 04, 2020. The manufacturer’s reason for the recall was “Possible out of specification condition which may allow a bead component to separate from an adjacent wire link. This condition may result in a discontinuous or open LINX device”. The link to the FDA recall is: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfres/res.cfm?id=163589.
Unfortunately, the literature is very scarce in reports about using the MR device after bariatric surgery and this specific complication of beads separation. There are only few articles published so far in the last 10 years[12-15].
The reason for not using the MR device after bariatric procedures could be multifactorial. From our own experience, we noticed a few reasons that may contribute to this condition. The first factor is the surgeons’ lack of experience with the MR device placement. In addition, the majority of bariatric surgeons are comfortable offering the RYGB as the treatment of choice for treating the reflux after SG and revising the RYGB if reflux occurs after RYGB. In addition, the majority of surgeons may not have been using this device in the general population; hence, they do not feel motivated or comfortable with adapting this technique. We can substantiate that theory in our group practice.
A second reason could be the delays and the denials of the insurance companies in covering the cost of this procedure, considering it to be experimental. The delays may take up to six months in some cases, as we have seen in our practice.
A third possibility is the incompatibility of the MR device with the latest version of the MRI (the 3 Tesla), creating fear for patients who are either receiving frequent MRIs or fearing the need for future MRI. In addition, allergy to nickel and foreign body sensation in some patients make the MR device not a suitable solution.
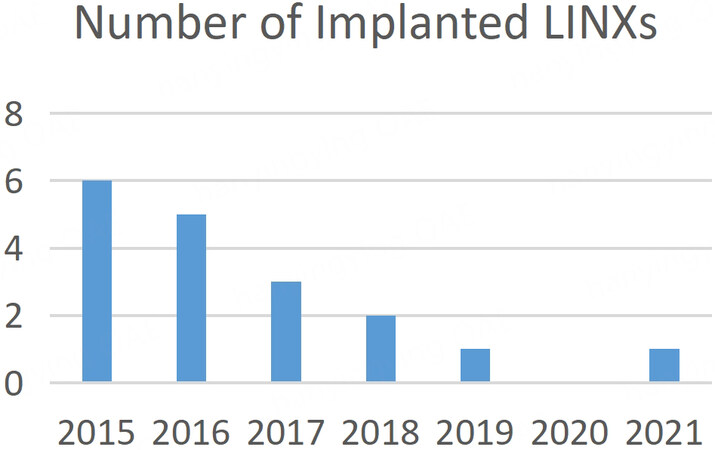
A fourth possibility is the early results of recurrent reflux after one year, along with the recent findings of broken magnetic ring device after 4 years, will further reduce the incentive to use this device in the future. Our own volume is an example of these possibilities for the decline in using the MR device, as depicted in Figure 8.
Our study is the first and only long-term study evaluating the MR device in controlling reflux after SG. It is also the first to report on a number of broken devices occurring after 3 years of implantation. The other studies on the use of the MR device in bariatric population usually evaluate the safety, feasibility and results of reflux control in the first or maybe the second year post implantation. This short period of follow-up is not enough to truly understand the effectiveness of the MR device in controlling reflux. We concluded this based on our finding of the majority of reflux recurrence and device complications occurring after 3 years. Our study results can be interpreted in two ways; on the one hand, it is disappointing when 81% (13/16) of patients are back on anti-reflux medication, and on the other hand, we can say that the MR device was able to reduce the reflux symptoms or resolve it in 44% of patients who were satisfied. It is interesting, however, to see that none of the patients with recurrent symptoms, who went back to using anti-reflux medication, wanted to undergo the RYGB conversion.
The limitation of our study is that it is retrospective and has a very small research base. Larger studies with similar or longer follow-up periods are needed to confirm or refute our findings before the final vote is cast on the fate of this device in managing reflux in the bariatric population especially if the company improves on the separation of the beads preventing this complication from occurring in the future.
In conclusion, the MR device is a valuable tool to treat reflux after SG when patients decline the conversion to RYGB, especially when the fundus of the stomach is not dilated enough for a fundoplication. The interpretation of the effectiveness of the device in controlling the reflux can vary depending on the definition of what is the meaning of a successful outcome. A specific degree of reduction in medication use can be considered as a successful outcome by one author, while a complete resolution of symptoms may be set as the ultimate goal to consider the device to be successful by another author.
DECLARATIONS
Authors’ contributionsMade substantial contributions to conception and design of the study, wrote the manuscript and performed data analysis and interpretation: Hawasli A
Performed data acquisition and data analysis, and provided administrative, technical, and material support: Hallowell S
Availability of data and materialsData is available upon request.
Financial support and sponsorshipNone.
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateThe hospital Institutional Review Board (IRB) deemed this study exempt as these patients were in the registry of the local Michigan Bariatric Surgery Collaborative (MBSC) and the national The Metabolic and Bariatric Surgery Accreditation and Quality Improvement Program (MBSAQIP) of the American College of Surgeons. Consent to participate from the patients was obtained.
Consent for publicationAll patients in this study had consented to the use of their data and intra- and post-operative images.
Copyright© The Author(s) 2023.
REFERENCES
1. Reynolds JL, Zehetner J, Bildzukewicz N, Katkhouda N, Dandekar G, Lipham JC. Magnetic sphincter augmentation with the LINX device for gastroesophageal reflux disease after U.S. food and drug administration Approval. Am Surg 2014;80:1034-8.
2. Desart K, Rossidis G, Michel M, Lux T, Ben-David K. Gastroesophageal reflux management with the LINX® system for gastroesophageal reflux disease following laparoscopic sleeve gastrectomy. J Gastrointest Surg 2015;19:1782-6.
3. Hawasli A, Bush A, Hare B, Meguid A, Thatimatla N, Szpunar S. Laparoscopic management of severe reflux after sleeve gastrectomy, in selected patients, without conversion to Roux-en-Y gastric bypass. J Laparoendosc Adv Surg Tech A 2015;25:631-5.
4. Hawasli A, Tarakji M, Tarboush M. Laparoscopic management of severe reflux after sleeve gastrectomy using the LINX® system: technique and one year follow up case report. Int J Surg Case Rep 2017;30:148-51.
5. Doulami G, Triantafyllou S, Natoudi M, et al. GERD-related questionnaires and obese population: can they really reflect the severity of the disease and the impact of GERD on quality of patient’s life? Obes Surg 2015;25:1882-5.
6. Mahawar KK, Jennings N, Balupuri S, Small PK. Sleeve gastrectomy and gastro-oesophageal reflux disease: a complex relationship. Obes Surg 2013;23:987-91.
7. Li JF, Lai DD, Lin ZH, Jiang TY, Zhang AM, Dai JF. Comparison of the long-term results of Roux-en-Y gastric bypass and sleeve gastrectomy for morbid obesity: a systematic review and meta-analysis of randomized and nonrandomized trials. Surg Laparosc Endosc Percutan Tech 2014;24:1-11.
8. Madalosso CA, Gurski RR, Callegari-Jacques SM, Navarini D, Thiesen V, Fornari F. The impact of gastric bypass on gastroesophageal reflux disease in patients with morbid obesity: a prospective study based on the Montreal Consensus. Ann Surg 2010;251:244-8.
9. Varban OA, Hawasli AA, Carlin AM, et al. Variation in utilization of acid-reducing medication at 1 year following bariatric surgery: results from the Michigan Bariatric Surgery Collaborative. Surg Obes Relat Dis 2015;11:222-8.
10. Zhang L, Tan WH, Chang R, Eagon JC. Perioperative risk and complications of revisional bariatric surgery compared to primary Roux-en-Y gastric bypass. Surg Endosc 2015;29:1316-20.
11. Hawasli A, Phillips A, Tarboush M. Laparoscopic management of reflux after Roux-en-Y gastric bypass using the LINX system and repair of hiatal hernia: a case report. Surg Obes Relat Dis 2016;12:e51-4.
12. Khaitan L, Hill M, Michel M, et al. Feasibility and efficacy of magnetic sphincter augmentation for the management of gastroesophageal reflux disease post-sleeve gastrectomy for obesity. Obes Surg 2023;33:387-96.
13. Muñoz-Largacha JA, Hess DT, Litle VR, Fernando HC. Lower esophageal magnetic sphincter augmentation for persistent reflux after Roux-en-Y gastric bypass. Obes Surg 2016;26:464-6.
14. Kuckelman JP, Phillips CJ, Derickson MJ, Faler BJ, Martin MJ. Esophageal magnetic sphincter augmentation as a novel approach to post-bariatric surgery gastroesophageal reflux disease. Obes Surg 2018;28:3080-6.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Hawasli A, Hallowell S. The effectiveness of the magnetic ring (LINX®) Device in managing reflux after sleeve gastrectomy; long term results: a seven-years' experience. Mini-invasive Surg 2023;7:4. http://dx.doi.org/10.20517/2574-1225.2022.64
AMA Style
Hawasli A, Hallowell S. The effectiveness of the magnetic ring (LINX®) Device in managing reflux after sleeve gastrectomy; long term results: a seven-years' experience. Mini-invasive Surgery. 2023; 7: 4. http://dx.doi.org/10.20517/2574-1225.2022.64
Chicago/Turabian Style
Hawasli, Abdelkader, Shoshana Hallowell. 2023. "The effectiveness of the magnetic ring (LINX®) Device in managing reflux after sleeve gastrectomy; long term results: a seven-years' experience" Mini-invasive Surgery. 7: 4. http://dx.doi.org/10.20517/2574-1225.2022.64
ACS Style
Hawasli, A.; Hallowell S. The effectiveness of the magnetic ring (LINX®) Device in managing reflux after sleeve gastrectomy; long term results: a seven-years' experience. Mini-invasive. Surg. 2023, 7, 4. http://dx.doi.org/10.20517/2574-1225.2022.64
About This Article
Special Issue
Copyright
Data & Comments
Data

 Cite This Article 23 clicks
Cite This Article 23 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.